WO2002102302A2 - Method of preparing and formulations including hydrolyzed jojoba protein - Google Patents
Method of preparing and formulations including hydrolyzed jojoba protein Download PDFInfo
- Publication number
- WO2002102302A2 WO2002102302A2 PCT/US2002/012898 US0212898W WO02102302A2 WO 2002102302 A2 WO2002102302 A2 WO 2002102302A2 US 0212898 W US0212898 W US 0212898W WO 02102302 A2 WO02102302 A2 WO 02102302A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- protein
- jojoba
- dispersion
- hydrolyzed
- molecular weight
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/0212—Face masks
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/0208—Tissues; Wipes; Patches
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/64—Proteins; Peptides; Derivatives or degradation products thereof
- A61K8/645—Proteins of vegetable origin; Derivatives or degradation products thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q1/00—Make-up preparations; Body powders; Preparations for removing make-up
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q17/00—Barrier preparations; Preparations brought into direct contact with the skin for affording protection against external influences, e.g. sunlight, X-rays or other harmful rays, corrosive materials, bacteria or insect stings
- A61Q17/04—Topical preparations for affording protection against sunlight or other radiation; Topical sun tanning preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/08—Anti-ageing preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/10—Washing or bathing preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/02—Preparations for cleaning the hair
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/06—Preparations for styling the hair, e.g. by temporary shaping or colouring
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/12—Preparations containing hair conditioners
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q9/00—Preparations for removing hair or for aiding hair removal
- A61Q9/02—Shaving preparations
Definitions
- the present invention is broadly concerned with hydrolyzed jojoba protein and uses
- cosmetic products such as shampoos, shampoo conditioners, hair styling gels, hair conditioners, hair reparatives, hair tonics, hair fixatives, hair mousses, bath and shower gels, liquid soaps, moisturizing sprays, makeup, pressed powder
- hydrolyzed j oj oba protein which is preferably in the form of a mixture of amino acids, peptides and/or protein fractions derived from the hydrolysis of naturally occurring jojoba
- Jojoba is a dioecious wind-pollinated shrub, reaching a height of 1-5 meters and having a long life span (100-200 years). Jojoba is cultivated mainly in Arizona, Northern Mexico, Argentina and Israel. Genetic variability in morphology, anatomy and physiology within the
- Leaves are xerophytic with a thick cuticle, sunken stomata. They contain special tissue with a
- Flowers are apetalous: the female ones are usually solitary, one per two nodes although flowers every node or in clusters are not rare. The male flowers are clustered.
- Flower buds form in the axiles of leaves solely on the new vegetative growth occurring during the warm seasons under favorable temperatures and water regime. New flower buds are dormant and will open only after a cool season with enough cold units for the fulfillment of their chilling requirements.
- Anthesis occurs in the spring when the soil and air temperature rise to above 15°C. Severe water stress prevents opening of flowers.
- the jojoba fruit is a capsule containing one to three dark brown seeds that normally range in their dry weight between 0.5-1.1 g and contains 44-56% wax. Fruits ripen during the spring and early summer
- the oil is extracted from jojoba seed by conventional screw pressing tecliniques, leaving a residual defatted dry material which is high in native jojoba protein (typically on the order of 25-35% by weight protein).
- Jojoba oil is a light yellow liquid at room temperature and is made up of straight-chain esters of mono-unsaturated long chain fatty acids and fatty alcohols and has an average total carbon chain length of 42 carbons.
- the product may be isomerized, hydrogenated, sulfurized, chlorinated or transesterified, and has a wide range of industrial uses, mainly in cosmetics in
- uses include pharmaceuticals and as extenders for plastics, printers inks, gear-oil additives and lubricants.
- the present invention is directed to a new form of jojoba protein, namely hydrolyzed jojoba protein and derivatives thereof, as well as uses of such protein products in cosmetic formulations .
- the preferred hydrolyzed j oj oba protein and derivatives thereof in accordance with the invention comprise a mixture of amino acids, peptides and/or protein fractions derived from the hydrolysis of the naturally occurring j oj oba protein.
- Such hydrolysis is preferably carried out enzymatically, but if desired acid hydrolysis can also be employed.
- the hydrolyzed jojoba protein products of the present invention are derived from the hydrolysis of defatted jojoba meal.
- Jojoba meal generally comprises about 25- 35% natural protein. This protein content is not made up entirely of one single or specific
- j oj oba as in all plants, there are two basic types of proteins, namely enzymes and storage proteins. Enzymes are produced to synthesize specific molecules or catalyze very specific reactions, and are found in very minute quantities in plants. On the other hand, storage proteins are produced in large quantities by seeds and are stored for utilization when the plant begins growing. These storage proteins provide the building blocks for the growth of new plants and are of course the predominant proteins in jojoba meal.
- Jojoba meal comprises a wide variety of other components including significant quantities of carbohydrate and fiber. Jojoba meal further comprises
- jojoba meal also contains other antinutritional factors such as trypsin inhibitor, polyphenols, bitter taste, nonnutritive protein and indigestible jojoba oil.
- defatted joj oba meal will comprise between about 30-75% by weight, more preferably between about 45-60% by weight, and most preferably between about 50-55% by weight carbohydrate.
- Jojoba meal will generally comprise between about 25-35% by weight protein and more preferably between about 28-30%) protein.
- Defatted jojoba meal will also comprise between about 12- 15 %> by weight simmondsin, less than about 2% by weight detectable
- the resultant product will contain the hydrolyzed j oj oba proteins, as well as the other ingredients noted above.
- the hydrolysis reaction has little or no effect upon the carbohydrate and other components of the starting meal.
- the hydrolyzed j oj oba protein of the invention typically includes a mixture having an amino acid, peptide and/or protein fragment molecular weight range of from about 75-
- the other ingredients of the meal are present along with the hydrolyzed jojoba proteins.
- membrane filter the hydrolysis products it is often desirable to membrane filter the hydrolysis products in order to segregate the peptides and/or protein fragments to achieve different
- a hydrolyzed jojoba protein product of relatively high molecular weight is provided, with a molecular weight range for the respective peptides and/or fragments therein of from about 1,000-5,000 and with an average molecular weight of from about 3,000-4,000.
- a lower molecular weight fraction (sometimes
- jojoba amino acid fraction is produced wherein the respective amino acids and
- peptides exhibit a molecular weight range of from about 75-1,000, with an average molecular
- hydrolyzed jojoba protein and derivatives thereof can be produced as a dry
- jojoba protein or derivatives at a level of from about 18-35% by weight, more preferably from about 23-27% by weight.
- "derivatives" of hydrolyzed jojoba protein refers to changes in the structure of the individual amino acids, peptides and/or protein fragments produced by amino acid addition, deletion, replacement, substitution and/or modifications; mutants produced by recombinant and/or DNA shuffling; quaternized species; and all other chemically synthe-
- One particularly preferred class of hydrolyzed jojoba protein derivatives is the lipid derivatives, especially those synthesized using C12-C22 fatty acids.
- hydrolyzed jojoba protein is intended to embrace and cover not only the amino acids, peptides and/or protein fractions derived from the hydrolysis of naturally occurring
- jojoba protein does not refer to one single or specific protein derived from jojoba, but rather, as discussed above, is intended to encompass a complex mixture of various proteins found in jojoba.
- humectants emollients, conditioners, thickeners, moisturizing agents, opacifiers, pearl agents, buffering agents, slip agents, feel agents, anti-static agents, acidifiers, preservatives, film formers, plasticizers, setting agents and suspending agents (usually, each of the foregoing ingredients
- the formulation of the invention comprise from about 10-95% by weight water, more preferably from about 20-75% by weight water.
- cosmetic products which can benefit from incorporation of the jojoba products of the invention are those selected from the group consisting of shampoos, shampoo conditioners, hair styling gels, hair conditioners, hair reparatives, hair tonics, hair fixatives, hair
- the shampoos and shampoo conditioners further comprise at least about 6% by weight detergent; the hairstyling gels further comprise a gel-forming polymer system; the hair conditioners further comprise at least about 0.3%) by weight cationic hair conditioner; the hair reparatives further comprise at least about 2% by weight cationic hair conditioner; the bath and shower gels further comprise at least about 6% by weight detergent; the hairstyling gels further comprise a gel-forming polymer system; the hair conditioners further comprise at least about 0.3%) by weight cationic hair conditioner; the hair reparatives further comprise at least about 2% by weight cationic hair conditioner; the bath and shower gels further
- the skin lotions and creams further comprise at least about 2% by weight of a cream former and emollients and/or oils; the sunscreen further comprising a sunblocking agent; and the shaving creams further comprising at least about 10%
- Figure 1 is a schematic flow diagram illustrating the steps involved in the preferred process for the production of hydrolyzed jojoba protein.
- This example describes a preferred procedure for the preparation of hydrolyzed jojoba protein, using solvent extracted defatted jojoba meal as a starting material.
- the meal is obtained from jojoba which has been conventionally press-treated with subsequent solvent extraction to
- the meal had a protein content of 30.2% by weight, 38.3% by weight dietary fiber, 1.7% by weight residual oil, ash of 4.3% by weight,
- the hydrolysis method is schematically illustrated in Fig. 1, and was carried out as
- step 14 the pH of the slurry was adjusted to 6.5 using lactic acid, whereupon a second protease treatment (step 16) was carried out.
- This treatment included addition of 10 pounds of a second protease enzyme (Flavorzyme, Novo) followed by agitation for 4 hours.
- 3 pounds of Dual Protease Enzyme (Enzyme Development Corp.) was dispersed in 1 gallon of tap water and added to the slurry followed by the addition of 10 pounds of Neutrase
- step 18 lactic acid was added to the slurry to lower the pH to 4.5, followed by the addition of 8 pounds sodium metabisulfite with agitation for 10 minutes.
- step 20 the slurry was heated to 160°F to deactivate all protease enzymes.
- the slurry containing hydrolyzed jojoba proteins was then processed in a rotary vacuum filter (step 22) to remove insolubles, and the filtrate was clarified by passage through a packed- house filter unit (step 24).
- the clarified filtrate from step 24 was then fed to a nanofiltration membrane system, in order to generate a permeate and a retentate having different molecular weight profiles (step 26)
- the membrane system was selected so that the lower molecular weight permeate proteins were generally below a molecular weight of 1,000 whereas the retentate proteins had a molecular weight of above
- the retentate fraction was first treated by the addition of preservatives (step 28), namely
- the permeate fraction was conventionally evaporated (step 40) to achieve a solids content
- aqueous liquid jojoba amino acid product 44 This liquid product contained about 4.5% by weight protein.
- amino acid product was a mixture of amino acids and peptides
- the jojoba amino acid products was analyzed to contain (wb) 0.28% aspartic acid, 0.13% threonine, 0.15% serine, 0.38% glutamic acid, 0.09% proline, 0.17% glycine, 0.17% alanine, 0.03% cysteine, 0.14% valine, 0.04% methionine, 0.09% isoleucine, 0.17% leucine,
- the two products developed using this process comprise amino acids, peptides and/or protein fragments derived from naturally occurring j oj oba protein, and that
- hydrolyzed jojoba protein and “hydrolyzed jojoba amino acid” is a
- a jojoba amino acid product is produced by acid hydrolysis of jojoba meal.
- 450 gallons of warm (160°F) water is transferred to a homomixer tank, followed by the addition of 2 gallons of concentrated HC1 (36%>). The mixture is stirred and 950
- the neutralized hydrolyzate is then clarified using a rotary vacuum filter to remove solid
- the clarified hydrolyzate is concentrated to the desired solids level (20-30%) in an evaporator.
- Preservatives 1% Germaben and 0.3%> Dowicil 200 are added.
- the product is then chilled at 34-35 °F for 16-24 hours. After chilling, the product is filtered using a packed-house filter unit to remove haziness.
- the product is then aged for 1-2 weeks, and a final filtration is carried out using a packed-house filter unit.
- the product is then packaged in suitable containers
- Example 3 quaternized derivatives of hydrolyzed joj oba protein or jojoba amino acid
- decylammonium chloride (Quab 342, 40% active) and 6 parts by weight of tap water are mixed in a separate container, followed by 1.7 parts by weight of 50% NaOH with agitation.
- Quab 342 Alternatives to the Quab 342 product are 3-cUoro-2-hydroxypropyl-N,N,N-dimethyloctadecylammonium chloride (Quab 426) or 3-chloro-2-hydroxypropyl-N,N,N-trimethylammonium chloride (Quat 188) to produce other derivatives.
- the solution in the separate container is then added to the pH-adjusted jojoba mixture, followed by agitation for 16-24 hours.
- the pH of the solution is then adjusted 4.4-4.6 using lactic acid (88%) solution.
- the product is then filtered using a packed-house filter unit to
- the finished product is laurdimonium hydroxypropyl hydrolyzed jojoba protein (or laurdimonium hydroxypropyl jojoba amino acid). If Quab 426 is used, the product
- jojoba amino acid If Quat 188 is used, the product is called hydroxypropyl trimonium hydrolyzed jojoba protein (or hydroxypropyl trimonium jojoba amino acid).
- Example 4 The following products were produced using the hydrolyzed jojoba protein products 39
- the product was prepared by placing an amount of distilled water in a primary tank and heating the water to 75 °C, whereupon the remaining ingredients of Phase A were added with mixing. The mixture was then cooled to 45 ° C and the ingredients of Phases B and C were added
- Distilled water was placed in a primary tanlc and the remaining ingredients of Phase A were added, followed by those ofPhases B and C, with mixing. pH was adjusted to 5.5-6.5 using
- the ultra shampoo was prepared using the same technique for the above-described hair shampoo.
- the moisturizing shampoo was prepared using the same technique for the above-
- the following ingredients were used to prepare the hand lotion.
- the distilled water was placed in a mixing tank along with glycerin, and the mixture was heated to 75 °C.
- the ingredients of Phase B were placed in a secondary tank and also heated to 75 °C.
- Phase B was then added to Phase A at 75 °C with good agitation.
- the mixture was then allowed to cool to 65 °C and the ingredient of Phase C was added, making sure that there were no lumps or powder remaining on the side of the tank.
- the ingredients of Phase D were added in the listed order.
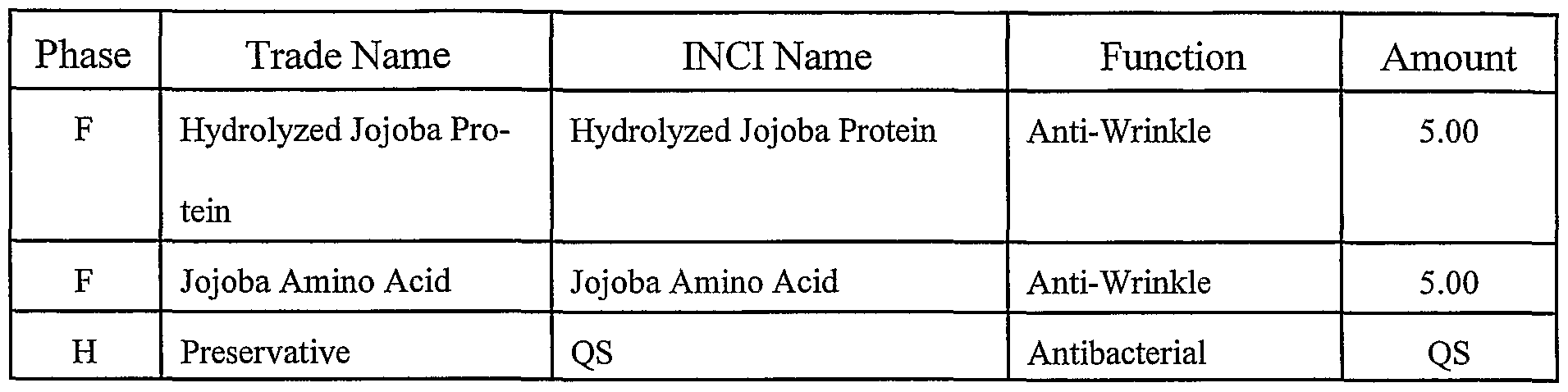
- the mixture was then cooled to 35 ° C and the j oj oba amino acid product 44 was added along with preservative.
- Phase B The ingredients of Phase B were placed in a secondary tank and also heated to
- Phase B ingredients were added to the Phase A ingredients at 75 °C with good agitation. The mixture was then allowed to cool and at 50-55 °C the ingredient of Phase C was
- Phase D The ingredients of Phase D were then added in the order listed. The mixture was then allowed to cool to 35 °C, whereupon the jojoba amino acid product and preservative were added.
- Phase B All of the ingredients in Phase B were weighed in another tank and heated to 75 ° C. Phase B was then added to Phase A with good agitation, and when the temperature reached 50-55 °C, the Skin-Flow-C wheat starch product was added,
- Phase A The ingredients of Phase A were heated to 75 °C, and the ingredients of Phase E were passed through a colloid mill with some propylene glycol and recirculated until the pigments were evenly dispersed.
- the colloid mill was rinsed with the Phase C ingredient and mixed using a lightnin mixer with heating to 75 °C.
- Phase B components were premixed and heated to 75 ° C, and added to the main batch.
- the Phase G ingredient was then sprinkled into the main batch premix Phase D with heating to 65 °C.
- the ingredients of Phase F were added to the main batch together with preservative and fragrance.
- Distilled water was metered into a primary tank and the glycerine was added with heating to 75 °C.
- the Phase B ingredients were added in order to a secondary tank and heated to 75 °C.
- Phase B ingredients were added to the Phase A ingredients with good agitation. When the temperature reached 50-55 °C, the Skin-Flow-C product was sifted into the batch, making sure there were no lumps or powder.
- the Phase D ingredients were added, and at 35 °C, the preservative was added with pH adjustment to 3.5-4.5.
- the distilled water was added to a primary tank followed by the ingredients of Phase A,
- Phase B ingredients were added in order to a secondary tank and
- Phase B was added to Phase A at 75° C with good agitation with continued mixing and cooling to 50-55 °C.
- the Skin-Flow-C product was added, making sure there were no lumps or powder remaining.
- the temperature reached 35 °C the jojoba amino acid and hydrolyzed jojoba protein were added, together with the preservative.
- the following ingredients were used to prepare the hair styling gel.
- the Carbomer was added in 25% water followed by mixing in of Phase A ingredients in order. Next, the Phase B ingredients were added with good agitation, followed by addition of preservative and fragrance. The pH of the product was 5.5. Professional Relaxer
- Phase A ingredients and Phase B ingredients were separately heated to 75 °C and
- the Phase C ingredient was then added at 50-55 °C.
- the product had a pH of 11.75-12.5.
- Phase A and Phase B ingredients were separately heated to 75 °C, and Phase B was added to Phase A with mixing. When the mixture reached 65° C, the Phase C ingredient was added with good mixing. Thereafter, the Phase D ingredients were added and the mixture was cooled to 35 °C, whereupon the Phase E ingredients were added along with the preservative.
Abstract
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2003504891A JP4229236B2 (en) | 2001-04-23 | 2002-04-23 | Method for preparing hydrolyzed jojoba protein and formulation comprising hydrolyzed jojoba protein |
| AU2002258979A AU2002258979A1 (en) | 2001-04-23 | 2002-04-23 | Method of preparing and formulations including hydrolyzed jojoba protein |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US09/841,552 | 2001-04-23 | ||
| US09/841,552 US6552171B2 (en) | 2001-04-23 | 2001-04-23 | Hydrolyzed jojoba protein |
| US09/841,544 | 2001-04-23 | ||
| US09/841,544 US6649177B2 (en) | 2001-04-23 | 2001-04-23 | Formulations including hydrolyzed jojoba protein |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2002102302A2 true WO2002102302A2 (en) | 2002-12-27 |
| WO2002102302A3 WO2002102302A3 (en) | 2003-03-13 |
Family
ID=27126265
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2002/012898 WO2002102302A2 (en) | 2001-04-23 | 2002-04-23 | Method of preparing and formulations including hydrolyzed jojoba protein |
Country Status (3)
| Country | Link |
|---|---|
| JP (1) | JP4229236B2 (en) |
| AU (1) | AU2002258979A1 (en) |
| WO (1) | WO2002102302A2 (en) |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1374848A1 (en) * | 2002-06-10 | 2004-01-02 | Neutrogena Corporation | Foaming clay cleanser composition |
| WO2008115165A2 (en) * | 2004-04-20 | 2008-09-25 | Mgp Ingredients, Inc. | Method of hydrolyzing rice protein concentrate with protease enzymes |
| CN106212453A (en) * | 2016-07-05 | 2016-12-14 | 南京科翼新材料有限公司 | A kind of glufosinate-ammoniumaqua aqua builder containing trimethylamino acid |
| US9586063B2 (en) | 2014-04-25 | 2017-03-07 | The Procter & Gamble Company | Method of inhibiting copper deposition on hair |
| US9642788B2 (en) | 2014-04-25 | 2017-05-09 | The Procter & Gamble Company | Shampoo composition comprising gel matrix and histidine |
| US9642787B2 (en) | 2014-04-25 | 2017-05-09 | The Procter & Gamble Company | Method of inhibiting copper deposition on hair |
| FR3092249A1 (en) * | 2019-02-06 | 2020-08-07 | Societe Industrielle Limousine D'application Biologique | COSMETIC ACTIVE INGREDIENT BASED ON SIMMONDSIA CHINENSIS SEED BREAD AND COSMETIC USES |
| WO2021037314A1 (en) * | 2019-08-31 | 2021-03-04 | Unifer International GmbH | Biostimulant agent for treating plants and/or plant seed |
| US10973744B2 (en) | 2016-06-30 | 2021-04-13 | The Procter And Gamble Company | Conditioner composition comprising a chelant |
| US11166894B2 (en) | 2016-06-30 | 2021-11-09 | The Procter And Gamble Company | Shampoo compositions comprising a chelant |
| US11246816B2 (en) | 2016-06-30 | 2022-02-15 | The Procter And Gamble Company | Shampoo compositions comprising a chelant |
| US11458085B2 (en) | 2016-06-30 | 2022-10-04 | The Procter And Gamble Company | Hair care compositions for calcium chelation |
| US11786447B2 (en) | 2016-06-30 | 2023-10-17 | The Procter & Gamble Company | Conditioner composition comprising a chelant |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5370996A (en) * | 1991-02-22 | 1994-12-06 | Calgene, Inc. | Fatty acyl reductases |
| US5403918A (en) * | 1991-02-22 | 1995-04-04 | Calgene, Inc. | Fatty acyl reductase |
| US5411879A (en) * | 1991-02-22 | 1995-05-02 | Calgene Inc. | Fatty acyl reductases |
| US5445947A (en) * | 1991-11-20 | 1995-08-29 | Calgene Inc. | Jojoba wax biosynthesis gene |
| US5679881A (en) * | 1991-11-20 | 1997-10-21 | Calgene, Inc. | Nucleic acid sequences encoding a plant cytoplasmic protein involved in fatty acyl-CoA metabolism |
| US5723747A (en) * | 1991-02-22 | 1998-03-03 | Calgene, Inc. | Wax esters in transformed plants |
| US6007823A (en) * | 1998-06-02 | 1999-12-28 | The United States Of America As Represented By The Secretary Of Agriculture | Simmondsin concentrate from jojoba |
-
2002
- 2002-04-23 AU AU2002258979A patent/AU2002258979A1/en not_active Abandoned
- 2002-04-23 WO PCT/US2002/012898 patent/WO2002102302A2/en active Application Filing
- 2002-04-23 JP JP2003504891A patent/JP4229236B2/en not_active Expired - Fee Related
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5370996A (en) * | 1991-02-22 | 1994-12-06 | Calgene, Inc. | Fatty acyl reductases |
| US5403918A (en) * | 1991-02-22 | 1995-04-04 | Calgene, Inc. | Fatty acyl reductase |
| US5411879A (en) * | 1991-02-22 | 1995-05-02 | Calgene Inc. | Fatty acyl reductases |
| US5723747A (en) * | 1991-02-22 | 1998-03-03 | Calgene, Inc. | Wax esters in transformed plants |
| US5445947A (en) * | 1991-11-20 | 1995-08-29 | Calgene Inc. | Jojoba wax biosynthesis gene |
| US5679881A (en) * | 1991-11-20 | 1997-10-21 | Calgene, Inc. | Nucleic acid sequences encoding a plant cytoplasmic protein involved in fatty acyl-CoA metabolism |
| US6007823A (en) * | 1998-06-02 | 1999-12-28 | The United States Of America As Represented By The Secretary Of Agriculture | Simmondsin concentrate from jojoba |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1374848A1 (en) * | 2002-06-10 | 2004-01-02 | Neutrogena Corporation | Foaming clay cleanser composition |
| WO2008115165A2 (en) * | 2004-04-20 | 2008-09-25 | Mgp Ingredients, Inc. | Method of hydrolyzing rice protein concentrate with protease enzymes |
| WO2008115165A3 (en) * | 2004-04-20 | 2009-05-22 | Mgp Ingredients Inc | Method of hydrolyzing rice protein concentrate with protease enzymes |
| US10835469B2 (en) | 2014-04-25 | 2020-11-17 | The Procter And Gamble Company | Method of inhibiting copper deposition on hair |
| US9586063B2 (en) | 2014-04-25 | 2017-03-07 | The Procter & Gamble Company | Method of inhibiting copper deposition on hair |
| US9642788B2 (en) | 2014-04-25 | 2017-05-09 | The Procter & Gamble Company | Shampoo composition comprising gel matrix and histidine |
| US9642787B2 (en) | 2014-04-25 | 2017-05-09 | The Procter & Gamble Company | Method of inhibiting copper deposition on hair |
| US11166894B2 (en) | 2016-06-30 | 2021-11-09 | The Procter And Gamble Company | Shampoo compositions comprising a chelant |
| US10973744B2 (en) | 2016-06-30 | 2021-04-13 | The Procter And Gamble Company | Conditioner composition comprising a chelant |
| US11246816B2 (en) | 2016-06-30 | 2022-02-15 | The Procter And Gamble Company | Shampoo compositions comprising a chelant |
| US11458085B2 (en) | 2016-06-30 | 2022-10-04 | The Procter And Gamble Company | Hair care compositions for calcium chelation |
| US11786447B2 (en) | 2016-06-30 | 2023-10-17 | The Procter & Gamble Company | Conditioner composition comprising a chelant |
| CN106212453B (en) * | 2016-07-05 | 2019-09-20 | 南京科翼新材料有限公司 | A kind of glufosinate-ammoniumaqua aqua builder of the acid containing trimethylamino |
| CN106212453A (en) * | 2016-07-05 | 2016-12-14 | 南京科翼新材料有限公司 | A kind of glufosinate-ammoniumaqua aqua builder containing trimethylamino acid |
| FR3092249A1 (en) * | 2019-02-06 | 2020-08-07 | Societe Industrielle Limousine D'application Biologique | COSMETIC ACTIVE INGREDIENT BASED ON SIMMONDSIA CHINENSIS SEED BREAD AND COSMETIC USES |
| WO2021037314A1 (en) * | 2019-08-31 | 2021-03-04 | Unifer International GmbH | Biostimulant agent for treating plants and/or plant seed |
| CN114501993A (en) * | 2019-08-31 | 2022-05-13 | 尤尼菲尔克斯国际有限公司 | Biostimulants for treating plants and/or plant seeds |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2004532895A (en) | 2004-10-28 |
| JP4229236B2 (en) | 2009-02-25 |
| WO2002102302A3 (en) | 2003-03-13 |
| AU2002258979A1 (en) | 2003-01-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR100613554B1 (en) | Cosmetics | |
| US6649177B2 (en) | Formulations including hydrolyzed jojoba protein | |
| JP4868646B2 (en) | Oat extract: purification, composition and method of use | |
| JP3040992B2 (en) | Food composition | |
| WO2002102302A2 (en) | Method of preparing and formulations including hydrolyzed jojoba protein | |
| US6955913B2 (en) | Unhydrolyzed jojoba protein products having high simmondsin concentration and methods of producing the same | |
| US6716599B2 (en) | Method of hydrolyzing defatted jojoba meal with protease enzymes | |
| TW201244637A (en) | Skin smoothing agent | |
| EP0977550B1 (en) | Use of at least one irvingia gabonensis extract in a cosmetic and/or pharmaceutical product | |
| JPS6368514A (en) | Cosmetic containing vegetable polypeptide derivative | |
| JP2001518910A (en) | Use of at least one protein fraction extracted from Hibiscus esculentus seed and cosmetic compositions comprising such a fraction | |
| WO2008115165A2 (en) | Method of hydrolyzing rice protein concentrate with protease enzymes | |
| JP2004331578A (en) | Cell activator | |
| JPH11308977A (en) | Beauty skin cosmetic composition | |
| JP2003104865A (en) | Skin care preparation | |
| JP2004331579A (en) | Production promoter of corium matrix | |
| JP2002284632A (en) | Extinction substance for superoxide anion extracted from sake lees as effective component | |
| JPH01249709A (en) | Cosmetic containing hen's egg polypeptide derivative | |
| JP2004026766A (en) | Skin care preparation for external use | |
| KR20200025834A (en) | Red sea cucumber-derived low salt peptide powder and process for producing the same | |
| JP2002020256A (en) | Skin care preparation | |
| JP3643785B2 (en) | Topical skin preparation | |
| KR20220080366A (en) | Preparing method of Tenebrio molitor extract for cosmetics | |
| JP3089602B2 (en) | Cosmetic raw material composition | |
| JP2021075504A (en) | Anti-inflammatory agent |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ OM PH PL PT RO RU SD SE SG SI SK SL TJ TM TN TR TT TZ UA UG UZ VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ OM PH PL PT RO RU SD SE SG SI SK SL TJ TM TN TR TT TZ UA UG UZ VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2003504891 Country of ref document: JP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| 122 | Ep: pct application non-entry in european phase |