WO1999022698A1 - Personal care compositions - Google Patents
Personal care compositions Download PDFInfo
- Publication number
- WO1999022698A1 WO1999022698A1 PCT/IB1998/001749 IB9801749W WO9922698A1 WO 1999022698 A1 WO1999022698 A1 WO 1999022698A1 IB 9801749 W IB9801749 W IB 9801749W WO 9922698 A1 WO9922698 A1 WO 9922698A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composition
- microemulsion
- organopolysiloxane
- silicone
- hair styling
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/0208—Tissues; Wipes; Patches
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/04—Dispersions; Emulsions
- A61K8/06—Emulsions
- A61K8/068—Microemulsions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/84—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions otherwise than those involving only carbon-carbon unsaturated bonds
- A61K8/89—Polysiloxanes
- A61K8/891—Polysiloxanes saturated, e.g. dimethicone, phenyl trimethicone, C24-C28 methicone or stearyl dimethicone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B82—NANOTECHNOLOGY
- B82Y—SPECIFIC USES OR APPLICATIONS OF NANOSTRUCTURES; MEASUREMENT OR ANALYSIS OF NANOSTRUCTURES; MANUFACTURE OR TREATMENT OF NANOSTRUCTURES
- B82Y5/00—Nanobiotechnology or nanomedicine, e.g. protein engineering or drug delivery
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/41—Particular ingredients further characterized by their size
- A61K2800/413—Nanosized, i.e. having sizes below 100 nm
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q1/00—Make-up preparations; Body powders; Preparations for removing make-up
- A61Q1/14—Preparations for removing make-up
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q17/00—Barrier preparations; Preparations brought into direct contact with the skin for affording protection against external influences, e.g. sunlight, X-rays or other harmful rays, corrosive materials, bacteria or insect stings
- A61Q17/005—Antimicrobial preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q17/00—Barrier preparations; Preparations brought into direct contact with the skin for affording protection against external influences, e.g. sunlight, X-rays or other harmful rays, corrosive materials, bacteria or insect stings
- A61Q17/04—Topical preparations for affording protection against sunlight or other radiation; Topical sun tanning preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/02—Preparations for cleaning the hair
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/06—Preparations for styling the hair, e.g. by temporary shaping or colouring
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/12—Preparations containing hair conditioners
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q9/00—Preparations for removing hair or for aiding hair removal
- A61Q9/02—Shaving preparations
Definitions
- the present invention relates to personal care compositions, especially hair styling compositions, containing silicone microemulsions, and corresponding applications of such compositions.
- the mvention further relates to stable, hair styling compositions containing a silicone microemulsion and lower monohydric alcohol.
- the desire to have the hair retain a particular style or shape is widely held.
- the most common methodology for accomplishing this is by spraying a composition, typically from a mechanical pump spray device or from a pressurized aerosol canister, to the hair.
- Other means of providing style or shaping to the hair are mousses, gels, lotions and the like.
- Such compositions provide temporary setting benefits and can usually be removed by water or by shampooing.
- the materials used in these types of hair styling compositions are generally resins, gums, and adhesive polymers which are capable of imparting style or shape to the hair. Many of these products also contain lower alcohols in order to obtain good films of the polymer in a short period of time.
- Stiff compositions tend to be brittle and break down under common stresses such as wind, brushing, combing. Stiff compositions also tend to feel and look unnatural.
- Sticky compositions overcome many of the foregoing disadvantages of stiff compositions, because sticky compositions tend to be more forgiving, i.e., flexible, under stress and allow for restyling of the hair.
- sticky compositions have the disadvantage of leaving the hair with a heavy, coated feel and with a limp and unattractive appearance.
- sticky compositions cause the hair to quickly become soiled from common contaminant sources such as dust, dirt, lint, sebum, etc.
- silicones including silicone emulsions and microemulsions
- Silicones tend to provide a desirably smooth or soft hair feel.
- silicone emulsions tend to be difficult to formulate in hair styling compositions.
- Hair styling compositions tend to be complex, requiring a number of ingredients for different purposes, with potential for incompatibilities.
- silicone emulsions tend to be unstable in compositions containing lower alcohols, resulting in phase separation of the composition.
- Other incompatibilities in the system e.g., polymer-polymer or polymer-surfactant interactions, can also result in phase separation. This phase separation is not only undesirable for visual esthetic reasons, but for performance reasons as well.
- hold and/or hair feel properties of the product tend to be negatively impacted.
- stable hair styling compositions providing these benefits and containing up to about 55% monohydric alcohol can be provided by employing a combination of a (i) non-silicone containing hair styling polymer, (ii) a silicone microemulsion substantially free of a combination of amino groups and hydroxyl groups and having a specific particle size, namely less than about 80nm, and (iii) certain solvent systems for the hair styling polymer.
- the present invention relates to hair styling compositions comprising: a) from about 0.01 % to about 20% of a non-silicone containing polymer suitable for hair styling; b) a organopolysiloxane microemulsion comprising: (i) a organopolysiloxane dispersed as particles in the microemulsion, the organopolysiloxane being substantially free of amino and hydroxyl groups in combination and having an average particle size of less than about 80 nanometers, and
- a surfactant system for dispersing the organopolysiloxane in the microemulsion wherein the amount of microemulsion is such that the personal care composition comprises from about 0.01% to about 10% of the organopolysiloxane; and c) a carrier comprising:
- a first solvent selected from the group consisting of water; water soluble organic solvents; organic solvents which are strongly to moderately strong in hydrogen-bonding parameter; and mixtures thereof; wherein the first solvent is other than Ci -C3 monohydric alcohol, C1-C3 ketone and C1-C3 ether; and
- compositions optionally, from about 0% to about 55% of a second solvent selected from the group consisting of C1 -C3 monohydric alcohols, C1-C3 ketones, C1-C3 ethers, and mixtures thereof.
- the composition comprises the second solvent which is preferably a C1 -C3 monohydric alcohol.
- Compositions of this type preferably comprise water as the first solvent.
- the present invention can comprise, consist of, or consist essentially of any of the required or optional ingredients and/or limitations described herein.
- compositions or components thereof so described are suitable for use in contact with human hair and the scalp and skin without undue toxicity, incompatibility, instability, allergic response, and the like.
- compatible means there is no marked phase separation, e.g., excessive cloudiness, layering or precipitation of the composition which negatively impacts the esthetic or functional properties of the composition in a significant manner.
- compositions are those wherein the mixture of essential components, namely the hair styling polymer, silicone microemulsion, and carrier is a substantially homogeneous solution or dispersion (preferably microdispersion), more preferably substantially clear to translucent in appearance.
- Preferred final compositions are those also characterized by these properties.
- compositions are those wherein the mixture of essential components provide a % transmittance of at least about 50% at a wavelength of 460 nm as determined by standard spectroscopy methods.
- Preferred final compositions are those also characterized by these properties.
- the composition of the present invention comprises a non-silicone containing hair styling polymer for providing stylability to the hair.
- Hair styling polymers possess adhesive properties such that they are capable of shaping or styling the hair, and should be removable by shampooing or rinsing the hair.
- One or more hair styling polymers may be used.
- the total amount of hair styling polymer is generally from about 0.01% to about 20%, preferably from about 0.1% to about 15%, more preferably from about 0.5% to about 10%>.
- a variety of hair styling polymers are suitable in the present invention. Particular polymers will be selected by the skilled artisan considering the solubility of the polymer in the composition and the ionicity of the composition.
- Suitable hair styling polymers are those which are soluble or dispersible in the carrier described herein in the ratios employed in the composition such that the polymer forms a substantially homogeneous solution or dispersion (preferably a microdispersion) with the carrier. Solubility/dispersibility is determined at ambient conditions of temperature and pressure (25°C and 101.3 kPa (1 Atm)). Solubility/dispersibility of the polymer should be determined after neutralization, if any.
- Suitable non-silicone-containing hair styling polymers include nonionic, anionic, cationic, and amphoteric polymers, and mixtures thereof.
- the non-silicone- containing hair styling polymers are preferably present in a combined amount of from about 0.01% to about 20%, more preferably from about 0.1% to about 15%, and most preferably from about 0.5% to about 10% by weight of composition.
- Suitable cationic polymers include Polyquaternium-4 (Celquat H-100; L200 - supplier National Starch); Polyquaternium-10 (Celquat SC-240C; SC-230 M - supplier National Starch); (UCARE polymer series - JR-125, JR-400, LR-400, LR- 30M, LK, supplier Amerchol ); Polyquaternium-11 (Gafquat 734; 755N - supplier ISP); Polyquaternium-16 (Luviquat FC 370; FC550; FC905; HM-552 supplier by BASF); PVP Dimethylaminoethylmethacrylate (Copolymer 845; 937; 958- ISP supplier); Vinyl Caprolactam/PVP/Dimethylaminoethyl Methacrylate copolymer (Gaffix VC-713; H2OLD EP-1 - supplier ISP); Chitosan (Kytamer L; Kytamer PC - supplier Amerchol
- Preferred cationic polymers are Polyquaternium-4; Polyquaternium-10; Polyquaternium-11 ; Polyquaternium-16; PVP/Dimethylaminoethylmethacrylate; Vinyl Caprolactam/PVP/Dimethylaminoethyl Methacrylate copolymer; and Chitosan.
- Suitable amphoteric polymers include Octylacrylmide/Acrylates/Butylaminoethyl Methacrylate Copolymer (Amphomer 28-4910, Amphomer LV-71 28-4971, Lovocryl-47 28-4947 - National Starch supplier), and Methacryloyl ethyl betaine/methacrylates copolymer (Diaformer series supplier Mitsubishi). Preferred are Octylacrylmide/Acrylates/Butylaminoethyl Methacrylate Copolymer.
- Especially preferred polymers for relatively low alcohol systems are those which are partially zwitterionic in that they always possess a positive charge over a broad range of pH but contain acidic groups which are only negatively charged at basic pH. Therefore the polymer is positively charged at lower pH and neutral (have both negative and positive charge) at higher pHs.
- the zwitterionic polymer may be selected from cellulose derivatives, wheat derivatives and chitin derivatives such as are known in the art.
- Nonlimiting examples of zwitterionic polymers useful herein include Polyquaternium-47 (Merquat 2001 - supplier Calgon (a zwitterionic copolymer of acrylic acid, methacryl ami do propyl trimethyl ammonium chloride, and methyl acrylate)); Carboxyl Butyl Chitosan (Chitolam NB/101 - marketed by Pilot Chemical Company, developed by Lamberti); and Dicarboxyethyl Chitosan (N-[(3'-hydroxy- 2',3'-dicarboxy)ethyl]-beta-D-(l,4)-glucosamine) (available from Amerchol as, e.g., CHITOLAM NB/101).
- Useful nonionic polymers include PVP or Polyvinylpyrrolidone (PVP K-15, K-30, K-60, K-90, K-120 - supplier ISP) (Luviskol K series 12, 17, 30, 60, 80, & 90 - supplier BASF); PVP/VA (PVP/VA series S-630; 735, 635, 535, 335, 235 - supplier ISP )(Luviskol VA); PVP/DMAPA acrylates copolymer (Styleze CC-10 - supplier ISP); PVP/V A/Vinyl Propionate copolymer (Luviskol VAP 343 E, VAP 343 I, VAP 343 PM - supplier BASF); Hydroxylethyl Cellulose (Cellosize HEC - supplier Amerchol); and Hydroxylpropyl Guar Gum (Jaguar HP series -8, -60, -105, -120 - supplier Rh ⁇ ne-Poulenc).
- Preferred nonionic polymers are PVP or Polyvinylpyrrolidone; PVP/VA; PVP/DMAPA acrylates copolymer; and Hydroxylpropyl Guar Gum.
- Anionic polymers suitable for use herein include VA/Crotonates/Vinyl Neodecanonate Copolymer (Resyn 28-2930 - National Starch supplier); Butyl Ester of PVM/MA (Gantrez A-425; ES-425; ES-435 - supplier ISP); Ethyl Ester of PVM/MA (Gantrez ES-225; SP-215 - supplier ISP); Acrylates/acrylamide copolymer (Luvimer 100P; Lumiver Low VOC, supplier BASF); Methacrylate Copolymer (Balance 0/55 - National Starch supplier); Vinyl Acetate/Crotonic Acid copolymer (Luviset CA 66 - supplier BASF); Isopropyl Ester of PVM/MA Copolymer (Gantrez ES-335 - supplier ISP); Acrylates Copolymer; Methacrylates/acrylates copolymer/amine salt (Diahold polymers - supplier Mitsubishi); 2-Butened
- Preferred anionic polymers are VA/Crotonates/Vinyl Neodecanonate Copolymer; Butyl Ester of PVM/MA; Ethyl Ester of PVM/MA; Acrylates/acrylamide copolymer; Methacrylate Copolymer; and Vinyl Acetate/Crotonic Acid copolymer.
- compositions of the present invention contain an organopolysiloxane microemulsion comprising polysiloxane particles dispersed in a suitable carrier (typicallly aqueous) with the aid of a surfactant.
- a suitable carrier typically aqueous
- the organopolysiloxane is substantially free of, preferably essentially free of, more preferably contains no, organopolysiloxane having both amino groups and hydroxyl groups.
- the microemulsions are preferably included in an amount such that the composition contains from about 0.01 to about 10% of the dispersed polysiloxane, more preferably about 0.05% to about 6%, most preferably about 0.1% to about 4%>.
- Organopolysiloxane microemulsions can be produced by the emulsion polymerization of organosiloxane having a low degree of polymerization in a solvent comprising water.
- the organopolysiloxane is stabilized in the microemulsion by a surfactant, e.g., a nonionic surfactant and an ionic surfactant.
- the average particle size of the emulsion after emulsion polymerization (corresponding to the organopolysiloxane in the emulsion) is less than about 80 nanometers (nm), preferably less than about 60 nm, more preferably less than about 40 nm.
- Particle size of a microemulsion can be determined by conventional methods, e.g., using a Leeds & Northrup Microtrac UPA particle sizer. Microemulsions having these particle sizes are more stable and have better external appearance than those having larger particle sizes. Furthermore, the degree of polymerization (DP) of the polysiloxane after emulsion polymerization is preferably in the range of from 3 to 5,000, more preferably in the range of from 10 to 3,000.
- the organopolysiloxane in the microemulsion can be a linear or branched chain siloxane fluid having a viscosity of about 20-3,000,000 mm ⁇ /s (cs), preferably 300-300,000 cs, more preferably 350-200,000 cs, at 25°C.
- Suitable organopolysiloxanes may contain the difunctional repeating "D" unit: wherein n is greater than 1 and Rl and R ⁇ are each independently Ci -C7 alkyl or phenyl.
- a mixture of siloxanes may be used.
- Exemplary siloxanes include polydimethylsiloxane, polydiethylsiloxane, polymethylethylsiloxane, polymethylphenylsiloxane, and polydiphenylsiloxane.
- Siloxane polymers with dimethylsiloxane "D" units are preferred from an economic standpoint.
- Rl and R ⁇ may independently be a functional group other than methyl, e.g., carboxyalkyl, haloalkyl, acrylate, acryloxy, acrylamide, vinyl or mercaptoalkyl.
- the siloxane may be terminated with hydroxy groups, alkoxy groups such as methoxy, ethoxy, and propoxy, or trimethylsiloxy groups, preferably hydroxy or trimethylsiloxy.
- the emulsion can be prepared by the emulsion polymerization process described in EP 459500 (published December 4, 1992), incorporated herein by reference.
- stable, oil free polysiloxane emulsions and microemulsions are prepared by mixing a cyclic siloxane, a nonionic surfactant, an ionic surfactant, water, and a condensation polymerization catalyst.
- the mixture is heated and agitated at polymerization reaction temperature until essentially all of the cyclic siloxane is reacted, and a stable, oil free emulsion or microemulsion is formed.
- the reaction mix, especially surfactant levels, and conditions are controlled in order to provide the desired organopolysiloxane particle size.
- the emulsions and microemulsions typically have a pH of about 3 to about 10 (e.g., 6-7.5), and contain about 10 to about 70% by weight siloxane polymer, preferably about 25 to about 60%), about 0%) to about 30% by weight nonionic surfactant, about 0%> to about 30% by weight ionic surfactant, preferably about 0%> to about 20%>, the balance being water.
- Preferred emulsions and methods of making them are further described in U.S. Patent Application Serial No. 08/929,721, filed on September 15, 1997 in the names of Ronald P. Gee and Judith M. Vincent, incorporated herein by reference in its entirety.
- Microemulsions can also be produced by the emulsion polymerization process described in EPA 0268982, published June 6, 1988, assigned to Toray, incorporated herein by reference in its entirety.
- the microemulsion is prepared by a process in which a crude emulsion, consisting of polysiloxane having a low degree of polymerization, a first surfactant (anionic, cationic, and nonionic surfactants), and water, is slowly dripped into an aqueous solution containing a catalytic quantity of a polymerization catalyst and a second surfactant which acts as an emulsifying agent (which may be the same as the first surfactant, however, the surfactants should be compatible in the reaction mixture considering the ionicity of the reaction mixture).
- a crude emulsion consisting of polysiloxane having a low degree of polymerization, a first surfactant (anionic, cationic, and nonionic surfactants), and water, is slowly dripped into
- reaction mix and conditions are controlled to provide the desired organopolysiloxane particle size. Therefore, a dropwise addition of the crude emulsion into the aqueous solution of catalyst and surfactant of 30 minutes or longer is preferred in order to produce microemulsions having smaller particle sizes.
- the quantity of surfactant used in the catalyst plus the surfactant aqueous solution is from about 5 to about 70 weight %, more preferably from about 25 to about 60 per 100 weight parts polysiloxane in the crude emulsion.
- nonionic surfactant can be used to prepare the microemulsion.
- exemplary types of nonionic surfactants include silicone polyethers, both grafted and linear block, ethoxylated fatty alcohols, ethoxylated alcohols, ethoxylated alkyl phenols, Isolaureth-6 (polyethylene glycol ether of branched chain aliphatic C12 containing alcohols having the formula Ci2H25(OCH2CH2)6OH), fatty acid alkanolamides, amine oxides, sorbitan derivatives (e.g., commercially available from ICI Americas, Inc., Wilmington, DE, under the tradenames SPAN and TWEEN), and propylene oxide-ethylene oxide block polymers (e.g., commercially available from BASF Corp., Parsippany, NJ under the trademark PLURONIC).
- Ionic surfactants useful in preparing the microemulsion include any conventional anionic surfactant such as sulfonic acids and their salt derivatives. Ionic surfactants also include any conventional cationic surfactant used in emulsion polymerization. Surfactants of these types are well known in the art and are commercially available from a number of sources. Specific examples of these surfactant types are also disclosed in the above referenced patent application Serial No. 08/929,721.
- the surfactant can be used in the form of a single type of surfactant (e.g., anionic, cationic or nonionic), or the surfactant can be used as a combination of two or more types provided that they are compatible with each other and the other components of the composition.
- Preferred combinations of surfactant types include the combination of two or more types of anionic surfactants, the combination of two or more types of nonionic surfactants, the combination of two or more types of cationic surfactants, the combination of two or more types of surfactants selected from both the anionic and nonionic surfactants; and the combination of two or more types of surfactants selected from both the cationic and nonionic surfactants.
- the catalyst employed in the emulsion polymerization may be any catalyst capable of polymerizing cyclic siloxanes in the presence of water, including condensation polymerization catalysts capable of cleaving siloxane bonds.
- Exemplary catalysts include strong acids and strong bases, ionic surfactants such as dodecylbenzenesulfonic acid, phase transfer catalysts, and ion exchange resins where a catalyst is formed in situ.
- a given surfactant may also serve as the polymerization catalyst (e.g., alkylbenzenesulfonic acids, or quaternary ammonium hydroxides or salt thereof may function as both a surfactant and the polymerization catalyst).
- a surfactant system, catalyst and resulting microemulsion suitable for use in the compositions of the present invention can be selected by the skilled artisan considering the ionicity of the composition. In general, these materials are selected such that the total composition will be compatible.
- the silicone micremulsion may contain a silicone polyether copolyol, such as described herein.
- the compositions hereof may contain a silicone polyether.
- a silicone polyether may be added prior to making the batch composition hereof.
- the polyether is not dispersible in the silicone microemulsion, it is preferably mixed in about an equal portion of water containing from 10-50% C1-C3 monohydric alcohol, preferably ethanol, prior to combination with the silicone microemulsion. This pre- mix is then added to the other ingredients of the composition which have preferably been pre-mixed.
- Organopolysiloxane microemulsions are available from a number of commercial sources.
- organopolysiloxane microemulsions are manufactured by Dow Corning of Midland, MI:
- composition contains a hair styling polymer which is an anionic acrylate polymer
- DC 2-1845 and DC 2-5791 are prefened microemulsions.
- the hair styling polymer is a cationic polymer comprising nitrogen
- the DC 2-8194, DC2-1281, and/or DC 2-5932 microemulsions are prefened.
- compositions of the invention also comprise a carrier for the hair styling polymer and the silicone microemulsion.
- Suitable carriers are those in which the hair styling polymer is soluble or dispersible, preferably soluble or microdispersible, and wherein the organopolysiloxane of the microemulsion is dispersible.
- Choice of an appropriate carrier will also depend on the particular end use and product form contemplated (e.g., the hair styling polymer to be used, and the product form, e.g., for hair styling compositions such as hair spray, mousse, tonic, lotion or gel).
- the carrier is preferably suitable for application to the hair.
- the carrier is present at from about 0.5% to about 99.5%, preferably from about 5% to about 99.5%, most preferably from about 50% to about 95%, of the composition.
- compositions of the present invention comprise one or more suitable solvents for the hair styling polymer.
- suitable solvents for the hair styling polymer are those which form a homogeneous solution or dispersion (preferably microdispersion) with the hair styling polymer in the weight ratios used in the composition.
- Prefened solvent systems are those which form a substantially clear to translucent solution or dispersion (preferably microdispersion) with the hair styling polymers in the weight ratios used in the composition.
- Prefened solvents include those selected from the group consisting of water; water soluble organic solvents; organic solvents which are strongly to moderately strong in hydrogen-bonding parameter; and mixtures thereof; wherein the solvent is other than Ci -C3 monohydric alcohol, C1 -C3 ketone and C1 -C3 ether.
- Water is a prefened solvent. At least about 3% to about 99%, preferably at least about 5% to about 98%, of this type of solvent is used in the composition.
- Exemplary water soluble organic solvents other than C1-C3 monohydric alcohols, ketones and ethers include propylene glycol, glycerine, phenoxyethanol, dipropylene glycol, sugars, and mixtures thereof.
- Solvents which are moderately strong to strong in hydrogen-bonding parameter other than C1-C3 monohydric alcohols, ketones and ethers include esters, ethers, ketones, glycol monoethers (moderately H-bonded) and alcohols, amines, acids, amides and aldehydes (strongly H-bonded).
- a description and examples of solvents of this type are disclosed in Polymer Handbook, 2d. Ed., J. Brandrup and E.H. Immergut, Editors, John Wiley & Sons, N.Y., 1975, Section IV, page 337 - 348 (Table 2).
- Prefened solvents of this type are dibutyl phthalate, propylene carbonate, propylene glycol monomethyl ether, methyl acetate, methyl proprionate and mixtures thereof.
- Propylene glycol monomethyl ether, methyl acetate, methyl proprionate and mixtures thereof are prefened; methyl acetate is most prefened.
- solvents suitable for use herein are water soluble, organic volatile solvents selected from C1-C3 monohydric alcohols, C1 -C3 ketones, C1 -C3 ethers, and mixtures thereof, monohydric alcohols being prefened.
- Prefened solvents of this type are methylal, ethanol, w-propanol, isopropanol, acetone and mixtures thereof. More prefened are ethanol, n-propanol, isopropanol, and mixtures thereof.
- the composition comprises about 40% or more of such solvents, at least about 5% of water, a water soluble organic solvent, and/or an organic solvent which is strongly to moderately strong in hydrogen-bonding parameter is used.
- the carrier comprises (i) a solvent selected from the group consisting of water; water soluble organic solvents; organic solvents which are strongly to moderately strong in hydrogen-bonding parameter; and mixtures thereof; wherein the solvent is other than C1-C3 monohydric alcohol, C ⁇ - C3 ketone and C1-C3 ether; (ii) a solvent selected from the groups consisting of C1 - C3 monohydric alcohols, C1-C3 ketones, C1-C3 ethers, and mixtures thereof; and (iii) mixtures thereof; preferably a mixture thereof.
- Especially prefened are a mixture of water and C ⁇ -C3 monohydric alcohol, e.g., water-ethanol or water- isopropanol-ethanol.
- Another particularly prefened solvent system comprises one or more of propylene glycol monomethyl ether, methyl acetate, and methyl proprionate, preferably methyl acetate, optionally with one or more of water or a C ⁇ - C3 monohydric alcohol.
- the carrier may include other solvents, e.g., hydrocarbons (such as isobutane, hexane, decene, acetone), halogenated hydrocarbons (such as Freon), linalool, volatile silicon derivatives, especially siloxanes (such as phenyl pentamethyl disiloxane, methoxypropyl heptamethyl cyclotetrasiloxane, chloropropyl pentamethyl disiloxane, hydroxypropyl pentamethyl disiloxane, octamethyl cyclotetrasiloxane, decamethyl cyclopentasiloxane), and mixtures thereof.
- hydrocarbons such as isobutane, hexane, decene, acetone
- halogenated hydrocarbons such as Freon
- linalool volatile silicon derivatives, especially siloxanes (such as phenyl pentamethyl disiloxane, methoxypropy
- Solvents used in admixture may be miscible or immiscible with each other. However, in the final composition such solvents should be compatible with each other and other components in the composition such that solids do not precipitate.
- Reduced “volatile organic compound” or “VOC” compositions may be desirable.
- VOC refers to those organic compounds that contain less than 12 carbon atoms or have a vapor pressure greater than about 0.1 mm of mercury.
- the composition may have, as initially applied, a total VOC content of about 55% or less (e.g., in prefened hairsprays), about 16% or less (e.g., in prefened mousses), or about 6% or less (e.g., in prefened gels).
- the VOC may be based on actual VOC content, or the VOC which is delivered upon initial dispensing from a package.
- the carrier may also comprise conventional components such as are known in the art suitable for a given product form.
- Optional Components are known in the art suitable for a given product form.
- compositions can contain a wide variety of other optional ingredients that are suitable for application to human hair, including among them any of the types of ingredients known in the art for use in hair care compositions, especially hair setting compositions like hair spray compositions, mousses, gels and tonics.
- such other adjuvants collectively comprise from about 0.05% to about 20% by weight, preferably from about 0.1% to about 10%, more preferably 0.05% to about 5% by weight of the compositions.
- Such conventional optional adjuvants include, but are not limited to, plasticizers, surfactants (which may be anionic, cationic, amphoteric or nonionic), neutralizing agents, propellants, hair conditioning agents (e.g., silicone fluids, fatty esters, fatty alcohols, long chain hydrocarbons, isobutene, cationic surfactants, etc.), emollients, lubricants and penetrants such as various lanolin compounds, vitamins, proteins, preservatives, dyes, tints, bleaches, reducing agents and other colorants, sunscreens, thickening agents (e.g., polymeric thickeners, such as xanthan gum), physiologically active compounds for treating the hair or skin (e.g., anti-dandruff actives, hair growth actives) and perfume.
- plasticizers e.g., surfactants (which may be anionic, cationic, amphoteric or nonionic), neutralizing agents, propellants, hair conditioning agents (e.
- compositions hereof may contain a plasticizer for the hair styling polymer.
- a plasticizer for the hair styling polymer Any plasticizer suitable for use in hair care products or for topical application to the hair or skin can be used.
- plasticizers are known in the art. These include glycerine, diisobutyl adipate, butyl stearate, propylene glycol, diethylene glycol, other glycols, tri-C2-Cg alkyl citrates, including triethyl citrate and analogs of triethyl citrate.
- Plasticizers are typically used at levels of from about 0.01% to about 200%, preferably from about 0.05% to about 100%, more preferably from about 0.1% to about 50%), by weight of the polymer.
- Surfactants are typically used at levels of from about 0.01% to about 200%, preferably from about 0.05% to about 100%, more preferably from about 0.1% to about 50%), by weight of the polymer.
- the hair styling compositions can contain one or more surfactants, e.g., for emulsifying hydrophobic components which may be present in the composition.
- Surfactants are prefened for use in mousse products. Generally, if used, such surfactants will be used at a total level of from about 0.01% to about 10%, preferably from about 0.01% to about 5% and more preferably from about 0.01% to about 3%, by weight of the composition.
- a wide variety of surfactants can be used, including anionic, cationic, amphoteric, and zwitterionic surfactants.
- Anionic surfactants include, for example: alkyl and alkenyl sulfates; alkyl and alkenyl ethoxylated sulfates; (preferably having an average degree of ethoxylation of 1 to 10), succinamate surfactants, such as alkylsulfosuccinamates and dialkyl esters of sulfosuccinic acid; neutralized fatty acid esters of isethionic acid; and alkyl and alkenyl sulfonates, including, for example, olefin sulfonates and beta-alkoxy alkane sulfonates.
- Prefened are alkyl and alkenyl sulfates and alkyl and alkenyl ethoxylated sulfates such as the sodium and ammonium salts of Ci 2-C ⁇ g sulfates and ethoxylated sulfates with a degree of ethoxylation of from 1 to about 6, preferably from 1 to about 4, e.g., lauryl sulfate and laureth (3.0) sulfate.
- Amphoteric surfactants include those which can be broadly described as derivatives of aliphatic secondary and tertiary amines in which the aliphatic radical can be straight chain or branched and wherein one of the aliphatic substituents contains from about 8 to about 18 carbon atoms and one contains an anionic water solubilizing group, e.g., carboxy, sulfonate, sulfate, phosphate, or phosphonate.
- anionic water solubilizing group e.g., carboxy, sulfonate, sulfate, phosphate, or phosphonate.
- Examples of compounds falling within this definition are sodium 3- dodecylaminopropionate, N-alkyltaurines such as the one prepared by reacting dodecylamine with sodium isethionate according to the teaching of U.S.
- Patent 2,658,072 N-higher alkyl aspartic acids such as those produced according to the teaching of U.S. Patent 2,438,091, and the products sold under the trade name "Miranol" and described in U.S. Patent 2,528,378.
- Others include alkyl, preferably C fi -C ⁇ and most preferably C ⁇ -C, -, amphoglycinates; alkyl, preferably C,--C .-> and most preferably C ⁇ -C, ⁇ , amphopropionates; and mixtures thereof.
- Suitable zwitterionic surfactants for use in the present compositions can be exemplified by those which can be broadly described as derivatives of aliphatic quaternary ammonium, phosphonium, and sulfonium compounds, in which the aliphatic radicals can be straight chain or branched, and wherein one of the aliphatic substituents contains from about 8 to 18 carbon atoms and one contains an anionic water-solubilizing group, e.g., carboxy, sulfonate, sulfate, phosphate, or phosphonate.
- a general formula for these compounds is:
- R2 Y (+) — CH 2 — R4 — Z (_) wherein R2 contains an alkyl, alkenyl, or hydroxy alkyl radical of from about 8 to about 18 carbon atoms, from 0 to about 10 ethylene oxide moieties and from 0 to 1 glyceryl moiety; Y is selected from the group consisting of nitrogen, phosphorus, and sulfur atoms; R3 is an alkyl or monohydroxyalkyl group containing 1 to about 3 carbon atoms; x is 1 when Y is sulfur or phosphorus, 1 or 2 when Y is nitrogen; R4 is an alkylene or hydroxyalkylene of from 1 to about 4 carbon atoms and Z is a radical selected from the group consisting of carboxylate, sulfonate, sulfate, phosphonate, and phosphate groups.
- Classes of zwitterionics include alkyl amino sulfonates, alkyl betaines, and alkyl amido betaines.
- Cationic surfactants useful in compositions of the present invention contain amino or quaternary ammonium hydrophilic moieties which are positively charged when dissolved in the aqueous composition of the present invention.
- Cationic surfactants among those useful herein are disclosed in the following documents, all incorporated by reference herein: M. C. Publishing Co., McCutcheon's, Detergents & Emulsifiers, (North American edition 1979); Schwartz, et al., Surface Active Agents, Their Chemistry and Technology, New York: Interscience Publishers, 1949; U.S. Pat. No. 3,155,591, Spotifyr, issued Nov. 3, 1964; U.S. Pat. No. 3,929,678, Laughlin, et al., issued Dec. 30, 1975; U.S. Pat. No. 3,959,461, Bailey, et al., issued May 25, 1976; and U.S. Pat. No. 4,387,090, Bolich, Jr., issued June 7, 1983.
- quaternary ammonium-containing cationic surfactant materials useful herein are those of the general formula:
- R ⁇ is an aliphatic group of from 1 to 22 carbon atoms, or an aromatic, aryl or alkylaryl group having from 12 to 22 carbon atoms
- R2 is an aliphatic group having from 1 to 22 carbon atoms
- R3 and R4 are each alkyl groups having from 1 to 3 carbon atoms
- X is an anion selected from halogen, acetate, phosphate, nitrate and alkylsulfate radicals.
- the aliphatic groups may contain, in addition to carbon and hydrogen atoms, ether linkages, and other groups such as amido groups.
- Other quaternary ammonium salts useful herein are diquaternary ammonium salts.
- Salts of primary, secondary and tertiary fatty amines are also suitable cationic surfactants for use herein.
- the alkyl groups of such amines preferably have from 12 to 22 carbon atoms, and may be substituted or unsubstituted.
- Secondary and tertiary amines are prefened, tertiary amines are particularly prefened.
- Such amines useful herein, include stearamido propyl dimethyl amine, diethyl amino ethyl stearamide, dimethyl stearamine, dimethyl soyamine, soyamine, myristyl amine, tridecyl amine, ethyl stearylamine, N-tallowpropane diamine, ethoxylated (5 moles E.O.) stearylamine, dihydroxy ethyl stearylamine, and arachidyl- behenylamine.
- Cationic amine surfactants included among those useful in the present invention are disclosed in U.S. Pat. No. 4,275,055, Nachtigal, et al., issued June 23, 1981 (incorporated by reference herein).
- Suitable cationic surfactant salts include the halogen, acetate, phosphate, nitrate, citrate, lactate and alkyl sulfate salts.
- Nonionic surfactants include polyethylene oxide condensates of alkyl phenols (preferably C6-C12 alkyl, with a degree of ethoxylation of about 1 to about 6), condensation products of ethylene oxide with the reaction product of propylene oxide and ethylene diamine, condensation products of aliphatic alcohols with ethylene oxide, long chain (i.e., typically C12-C22) tertiary amine oxides, long chain tertiary phosphine oxides, dialkyl sulfoxides containing one long chain alkyl or hydroxy alkyl radical and one short chain (preferably C1 -C3) radical, silicone copolyols, and C1-C4 alkanol amides of acids having a Cg ⁇ C22 ac yl moiety.
- alkyl phenols preferably C6-C12 alkyl, with a degree of ethoxylation of about 1 to about 6
- Prefened nonionic surfactants are C1 -C4 alkanol amides of acids having a Cg-C22 acyl moiety, polyoxyethylene glycol stearyl ethers, and mixtures thereof. Specific examples which are prefened are Lauramide DEA, Steareth-21, Steareth-2, and Na Cocoyl Isethionate..
- Hair styling polymers which have acidic functionalities, such as carboxyl groups, are preferably used in at least partially neutralized form to promote solubility/dispersibility of the polymer.
- use of the neutralized form aids in the ability of the dried hair styling compositions to be removed from the hair by shampooing.
- the degree of neutralization must balance shampoo removability versus humidity resistance. Neutralization levels in excess of what is required for shampoo removability will result in excessively sticky products that will not hold as well in high humidity.
- available acidic monomers When available acidic monomers are neutralized, it is prefened that from about 5% to 60%, more preferably from about 10% to about 40%), and even more preferably from about 12%> to about 30%> of the polymer (on a total polymer weight basis) be neutralized.
- the optimal level of neutralization for a specific polymer will depend on the polarity of the monomers selected, the specific ratios of the monomers to each other, and the percentage of acidic monomers.
- the level of base needed to neutralize the acid groups in a polymer for a specific % neutralization of the polymer may be calculated from the following equation:
- Any conventionally used base including organic or inorganic (metallic or other) bases, can be used for neutralization of the polymers.
- Metallic bases are particularly useful in the present compositions.
- Hydroxides, where the cation is ammonium, an alkali metal or an alkaline earth metal, are suitable neutralizers for use in the present compositions.
- Prefened inorganic neutralizing agents for use in the compositions of the present invention are potassium hydroxide and sodium hydroxide.
- Suitable neutralizing agents which may be included in the hair styling compositions of the present invention include amines, especially amino alcohols such as 2-amino-2 -methyl- 1,3-propanediol (AMPD), 2-amino-2- ethyl-l,3-propanediol (AEPD), 2-mino-2-methyl-l-propanol (AMP), 2-amino-l- butanol (AB), monoethanolamine (MEA), diethanolamine (DEA), triethanolamine (TEA), monoisopropanolamine (MIPA), diisopropanolamine (DIPA), triisopropanolamine (TIP A), dimethyl laurylamine (DML), dimethyl myristalamine (DMM) and dimethyl stearamine (DMS).
- AMPD 2-amino-2 -methyl- 1,3-propanediol
- AEPD 2-amino-2- ethyl-l,3-propanediol
- Polymers having basic functionalities, e.g., amino groups, are preferably at least partially neutralized with an acid, e.g., hydrochloric acid.
- Neutralization can be accomplished by techniques well known in the art, and before or after polymerization of the monomers comprising the hair styling polymer. d) Hair Conditioning Polymers
- compositions of the invention may include a hair conditioning polymer for purposes of improved wet combing, dry combing and/or improved manageability (e.g., frizz or static control). Hair conditioning polymers are typically used at a level of from about 0,001% to about 6%, more preferably from about 0.01%> to about 5% of the composition.
- Cationic and zwitterionic hair conditioning polymers are prefened.
- Suitable hair conditioning polymers include cationic polymers having a weight average molecular weight of from about 5,000 to about 10 million, and will generally have cationic nitrogen-containing moieties such as quaternary ammonium or cationic amino moieties, and mixtures thereof.
- Cationic charge density should be at least about 0.1 meq/gram, preferably less than about 3.0 meq/gram, which can be determined according to the well known Kjeldahl Method. Those skilled in the art will recognize that the charge density of amino-containing polymers can vary depending upon pH and the isoelectric point of the amino groups. The charge density should be within the above limits at the pH of intended use. Any anionic counterions can be utilized for the cationic polymers so long as they are compatible.
- the cationic nitrogen-containing moiety will be present generally as a substituent, on a fraction of the total monomer units of the cationic hair conditioning polymers.
- the cationic polymer can comprise copolymers, terpolymers, etc. of quaternary ammonium or cationic amine-substituted monomer units and other non- cationic units refened to herein as spacer monomer units.
- Such polymers are known in the art, and a variety can be found in International Cosmetic Ingredient Dictionary, Sixth Edition, 1995, which is incorporated by reference herein in its entirety.
- Suitable cationic polymers include, for example, copolymers of vinyl monomers having cationic amine or quaternary ammonium functionalities with water soluble spacer monomers such as acrylamide, methacrylamide, alkyl and dialkyl acrylamides, alkyl and dialkyl methacrylamides, alkyl acrylate, alkyl methacrylate, vinyl caprolactone, and vinyl pynolidone.
- the alkyl and dialkyl substituted monomers preferably have C1-C7 alkyl groups, more preferably C1 -C3 alkyl groups.
- Suitable spacer monomers include vinyl esters, vinyl alcohol (made by hydrolysis of poly- vinyl acetate), maleic anhydride, propylene glycol, and ethylene glycol.
- the cationic polymers are described in detail in U.S. Patent 4,733,677 which is hereby incorporated by reference to further describe the cationic polymers used for conditioning purposes.
- the cationic amines can be primary, secondary, or tertiary amines, depending upon the particular species and the pH of the composition. In general, secondary and tertiary amines, especially tertiary amines, are prefened.
- the cationic polymers can comprise mixtures of monomer units derived from amine- and/or quaternary ammonium-substituted monomer and/or compatible spacer monomers.
- cationic polymers that can be used include polysaccharide polymers, such as cationic cellulose derivatives, cationic starch derivative, and cationic guar gum derivatives.
- Other materials include quaternary nitrogen-containing cellulose ethers as described in U.S. Patent 3,962,418, and copolymers of etherified cellulose and starch as described in U.S. Patent 3,958,581, which descriptions are incorporated herein by reference.
- the pH of the zwitterion is preferably adjusted to that of the neutralized hair styling polymer prior to combination therewith.
- Neutralization may be achieved by conventional methods using pH- adjusting agents such as are known in the art.
- a propellant which consists of one or more of the conventionally-known aerosol propellants can be used to propel the compositions.
- a suitable propellant for use can be any gas conventionally used for aerosol containers, preferably a liquifiable gas.
- Suitable propellants for use are volatile hydrocarbon propellants which can include liquified lower hydrocarbons of 3 to 4 carbon atoms such as propane, butane and isobutane.
- propellants are hydro fluorocarbons such as 1,2-difluoroethane (Hydrofluorocarbon 152A) supplied as Dymel 152 A by DuPont.
- hydro fluorocarbons such as 1,2-difluoroethane (Hydrofluorocarbon 152A) supplied as Dymel 152 A by DuPont.
- propellants are dimethylether, nitrogen, carbon dioxide, nitrous oxide, and atmospheric gas.
- the selection of appropriate hydrocarbons is made to provide a stable system giving the desired spray/foam quality.
- the aerosol propellant may be mixed with the present hair styling compositions and the amount of propellant to be mixed is governed by normal factors well known in the aerosol art.
- the level of propellant is from about 1% to about 60% by weight of the total composition.
- the propellant level is from about 10% to about 60%> by weight of the total composition, preferably from about 15%> to about 50% by weight of the total composition.
- the level of propellant is generally from about 1% to about 30% and more preferably from about 4% to about 15% by weight of the total composition.
- pressurized aerosol dispensers can be used where the propellant is separated from contact with the hair styling composition such as a two compartment can of the type sold under the tradename SEPRO from American National Can Corp.
- Suitable aerosol dispensers are those characterized by the propellant being compressed air which can be filled into the dispenser by means of a pump or equivalent device prior to use. Such dispensers are described in U.S. Patents 4,077,441, March 7, 1978, Olofsson and 4,850,577, July 25, 1989, TerStege, both incorporated by reference herein, and in U.S. Serial No. 07/839,648, Gosselin et al., filed February 21, 1992, also incorporated by reference herein. Compressed air aerosol containers suitable for use herein are also those previously marketed by The Procter & Gamble Company under their tradename VIDAL SASSOON AIRSPRAY ® hair sprays.
- non-aerosol foams may also be mixed with the present hair styling composition such that the final composition is dispensable as a stable foam.
- a composition is "dispensable as a stable foam” when it produces a foam when dispensed from a package or container which is either pressurized or equipped with an air or gas mixing device like the F2 non-aerosol foamer described in U.S. Patents 5,271,530; 5,337,929; and 5,443,569; all of which are herein incorporated by reference.
- compositions of the present invention may contain a silicone polyether suitable for stabilizing the organopolysiloxane microemulsion.
- the silicone polyether is especially prefened for relatively high alcohol compositions (e.g., at least about 40% monohydric alcohol).
- the silicone polyether comprises a polymeric portion comprising repeating organosiloxane units, and a polymeric portion comprising repeating alkylene oxide units (i.e., a silicone-polyoxyalkylene copolymer).
- Suitable silicone polyethers are those which are surface active in the carrier employed in the compositions of the invention. As will be understood in the art, the surface activity of the silicone polyether will depend on the molecular weight of the polymeric portion comprising repeating organosiloxane units.
- the silicone polyether is used in an amount effective to stabilize the microemulsion, preferably from about 0.02% to about 7%, more preferably about 0.05% to about 5%, of the total composition.
- the silicone polyether may be a silicone - linear polyoxyalkylene block copolymer (wherein the polymeric backbone comprises silicone blocks and polyoxyalkylene blocks, optionally having grafts), a silicone - graft polyoxyalkylene copolymer (wherein the polymeric backbone comprises silicone blocks and the polyoxyalkylene blocks are present as grafts rather than in the backbone), or a mixture thereof. Linear copolymers are prefened.
- Silicone linear block polyethers particularly prefened for relatively high alcohol compositions (e.g., at least about 40% monohydric alcohol) include those having the formula (I):
- M' is a monofunctional unit R2R'SiO 2;
- D is a difimctional unit R2SiO2/2;
- D' is a difimctional unit RR'Si ⁇ 2/2;
- R is independently H, Ci - Cg alkyl, or aryl, preferably H or C ⁇ - C4 alkyl, more preferably CH3;
- R' is independently, an oxyalkylene containing moiety, H, or CH3; b is an integer of from about 10 to about 1000, preferably about 10 to about 500, more preferably about 20 to about 200; and c is an integer of from 0 to about 100, preferably 0 to about 50, more preferably c is 0, provided that when c is 0, at least one M' contains an oxyalkylene moiety.
- R' in structure (I) are those having the formula:
- R" is a divalent radical for connecting the oxyalkylene portion of moiety R' to the siloxane backbone, preferably - (C m H2 m ) -, wherein m is an integer of from 2 to 8, preferably from 2-6, more preferably from 3-6;
- R'" is a terminating radical for the oxyalkylene portion of the moiety R', e.g., H, hydroxyl, Ci - C ⁇ alkyl, aryl, alkoxy (e.g., Ci - C ⁇ ) or acyloxy (e.g., Ci - C ⁇ ), preferably hydroxyl;
- n is an integer of from 2 to 4, preferably 2 to 3 (i.e., the oxyalkylene group may contain ethylene oxide, propylene oxide and/or butylene oxide units); and y is 1 or greater, wherein the total y from all the oxyalkylene units in the copolymer is 10 or greater.
- the oxyalkylene moiety of R' may be a random copolymer, a block copolymer or a mixture thereof.
- Prefened R' groups in structure (I) are those wherein the oxyalkylene units are selected from ethylene oxide units (EO), propylene oxide units (PO), and mixtures thereof. More prefened are those wherein the oxyalkylene units have an ethylene oxide unit (EO) to propylene oxide unit (PO) ratio of EO ⁇ -lO ⁇ PO ⁇ -100' mor e preferably E ⁇ 2 ⁇ -7 ⁇ P ⁇ 20-70> m °st preferably EO30-70PO3O-7O' based on the total oxyalkylene in the silicone polyether.
- EO ethylene oxide unit
- PO propylene oxide unit
- Particularly prefened silicone polyethers for relatively high alcohol compositions are those having the formula:
- n is as defined above, x is independently an integer of 1 or greater, a and b independently are an integer of from about 15 to about 30, and w is an integer of from about 20 to about 200, preferably from about 30 to about 200.
- silicone polyethers are commercially available from Goldschmidt Chemical Company under the tradename TEGOPREN 5830.
- Prefened silicone graft polyethers for use herein are those having the formula (II):
- M is a mono functional unit R3SiO ⁇ /2 >
- D is a difimctional unit R2SiO2/2,
- D' is a difimctional unit RR'Si ⁇ 2/2 >
- R is independently H, Ci - C6 alkyl, or aryl, preferably H or Ci - C4 alkyl, more preferably CH3,
- R' is an oxyalkylene containing moiety
- b is an integer of from about 10 to about 1000, preferably about 100 to about 500
- c is an integer of from 1 to about 100, preferably 1 to about 50.
- Prefened R' are those having the formula
- R" is a divalent radical for connecting the oxyalkylene portion of moiety R' to the siloxane backbone, preferably - (C m H2 m ) -, wherein m is an integer of from 2 to 8, preferably 2-6, more preferably 3-6;
- R"' is a terminating radical for the oxyalkylene portion of moiety R', e.g., H, hydroxyl, Ci - C ⁇ alkyl, aryl, alkoxy (e.g., Ci - C ⁇ ) or acyloxy (e.g., C ⁇ - C ⁇ ), preferably hydroxy or acyloxy, more preferably hydroxyl; n is an integer of from 2 to 4, preferably 2 to 3 (i.e., the oxyalkylene group may contain ethylene oxide, propylene oxide and/or butylene oxide units); and y is 1 or greater.
- the oxyalkylene moiety of R' may be a random copolymer, a block copolymer or a mixture thereof.
- Prefened R' groups are those wherein the oxyalkylene units are selected from ethylene oxide units (EO), propylene oxide units (PO), and mixtures thereof. More prefened are those wherein the oxyalkylene units have an ethylene oxide unit (EO) to propylene oxide unit (PO) ratio of EO1 Q. lO ⁇ PO ⁇ -100' more preferably EO10-30 ⁇ 01-30' based on the total oxyalkylene in the silicone polyether.
- Table 1 shows some representative silicone graft polyethers:
- Silicone Polyethers A and B contain less than 20 D units and less than 5 D' units.
- Silicone Polyethers C-G contain from 100-200 D units and 10-30 D' units.
- Hydrophihc lipophihc balance is determined by calculating the weight percent of EO and dividing this value by five.
- Silicone polyethers of this type are further described in the above referenced patent application Serial No. 08/929,721.
- Siloxane-oxyalkylene copolymers i.e., silicone polyethers
- silicone polyethers can be prepared according to methods generally described in the standard text on silicone chemistry entitled “Chemistry and Technology of Silicones,” by Walter Noll, Academic Press Inc., Orlando, Florida, (1968), on pages 373-376. Silicone polyethers are also available from a number of commercial sources such as:
- silicone polyethers are available as SF-1188 offered by General Electric of Waterford, N.Y., and KF353A offered by Shin Etsu Silicones of America of Tonance, Ca.. Additional silicone polyethers are described in U.S. Patent 4,871,529, incorporated herein by reference. g) Other optional components
- compositions of the invention may contain a variety of other ingredients such as are conventionally used in a given product form.
- the compositions hereof may, for example, be in the form of a hairspray, mousse, gel, lotion, cream, pomade, spray-on product such as spray-on gel, heat protectant spray, volumizing spray, spritz, hair tonic, and the like.
- the compositions may be aerosol or non-aerosol.
- Such compositions are described, for example in California Code of Regulations, Regulation for Reducing Volatile Organic Compound Emissions from Consumer Products, Amendment 2, Consumer Products, Sections 94507-94717, Title 17, filed September 19, 1991 and effective October 21, 1991; and in Formulation and Function of Cosmetics, J. S. Jellinek, Wiley Interscience (1970), each incorporated herein by reference.
- the hair styling compositions of the present invention can be made using conventional formulation and mixing techniques.
- the hair styling polymer and the solvent are mixed to provide a homogeneous mixture. Any other ingredients are then added and mixed to yield the final composition. If the polymer is neutralized, the neutralizer is preferably added prior to addition of other ingredients.
- the composition is packaged in conventional mechanical pump spray devices, or alternatively, in the case of aerosol sprays products, the composition is packaged in conventional aerosol canisters along with an appropriate propellant system (also applicable for mousses).
- propellant system also applicable for mousses.
- Other hair styling compositions including tonics, lotions, and gels, are typically packaged in a conventional bottle or tube.
- compositions of the present invention comprise a silicone microemulsion
- water helps to maintain the stability of the silicone microemulsions in the composition.
- the amount of water which is preferably present depends on the type of hair styling polymer and the presence and type of silicone copolyol. If the composition does not contain a silicone copolyol, at least about 30% water is preferably present. Where the composition contains a silicone copolyol having structure (I) (e.g., Tegopren), at least about 3% water is preferably present. When the composition contains other dimethicone copolyols at least about 25% water is preferably present.
- I silicone copolyol having structure
- silicone microemulsion is typically added after any thickeners or surfactants.
- compositions of the present invention are used in conventional ways to provide the hair care benefits of the present invention. Such methods generally involve application of an effective amount of the product to dry, slightly damp, or wet hair before and/or after the hair is dried and ananged to a desired style.
- Application of the product is normally effected by spraying or atomizing the product using an appropriate device, e.g. a mechanical pump spray, a pressurized aerosol container, or other appropriate means.
- Other hair styling compositions including tonics, lotions, and gels, are typically dispensed from a conventional bottle or tube, and applied directly to the hair or first dispensed to the hand and then to the hair. The composition is then dried or allowed to dry.
- effective amount is meant an amount sufficient to provide the hair hold and style benefits desired. In general, from about 0.5g to about 30g of product is applied to the hair, depending upon the particular product formulation, dispenser type, length of hair, and type of hair style.
- compositions are also useful in other applications where the benefits of the composition hereof may be realized.
- other applications in the personal care area or household care area may benefit from a composition containing a polymer having hair styling properties, along with the silicone microemulsion.
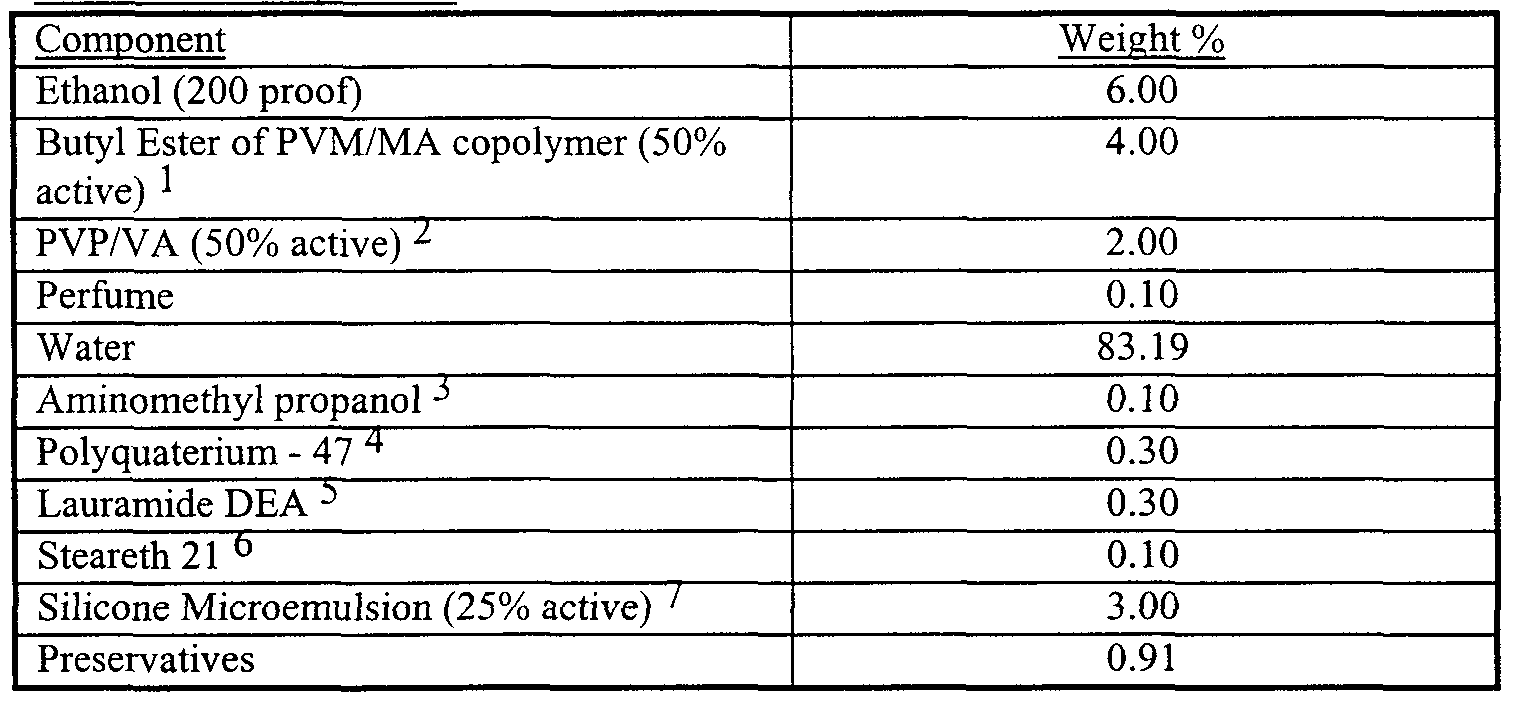
- Example I The following is a gel composition representative of the present invention.
- DC- 1845 - Silicone microemulsion from Dow Corning with a particle size of 33 nm, an aniomc/nonionic surfactant system, and a silicone with an internal phase viscosity 70,000 cps
- a premix is prepared by completely dissolving the PVP/VA in 30% of the batch water (100° F) with vigorous agitation In the following order, under continued agitation, add the Diethylene glycol, the Isosteareth-20, the perfume, the Glycerin, the ethanol and the preservatives.
- the final mix is prepared under vigorous agitation by dissolving the Carbomer in 50% of the batch water (120° F) until the carbomer is fully hydrated With mild agitation, add T ⁇ ethanol amine to carbomer/water, allowing gelation to occur With continued mild agitation, add the premix and the silicone microemulsion (premixed with the remaining water) to the mix.
- Example II The following is a gel composition representative of the present invention:
- DC-2-5932 silicone microemulsion from Dow Coming with a particle size of 24 nm, a cationic surfactant system, and a silicone with an internal phase visocosity 1,200 cps.
- a premix is prepared by adding the Polyquatern ⁇ um-16 to 60% of the water under moderate agitation. Next with vigorous agitation, add the Benzophenone, the Ethanol, the Perfume, and the preservatives.
- the final mix is prepared by addmg the hydroxypropyl guar to 30% of the water, mixing vigorously until the guar hydrates completely Next, add the premix with gentle agitation. Then add the silicone microemulsion (premixed with the remaining water) to the mix with mild agitation.
- the final mix is prepared under vigorous agitation by dissolving the HEC m 50% of the batch water (155° F) until the HEC is fully hydrated With mild agitation, add premix and then the silicone microemulsion (premixed with the remaining water)
- Example IV The following is a mousse composition representative of the present mvention Concentrate composition
- DC-2-5932 silicone microemulsion from Dow Coming with a particle size of 24 nm, a cationic surfactant system, and a silicone with an internal phase viscosity 1,200 cps
- the first premix is prepared by adding the ethanol to V_ of the water (save remaining l ⁇ for premixing Silicone Microemulsion, preservatives and surfactants) Next, add the Celquat H- 100 and mix until uniformly dissolved in ethanol water Add the Gafquat-755N and mix until uniformly dissolved With vigorous agitation, add the perfume and the glycerm
- the second premix is prepared by dissolving the surfactants (Lauramide DEA, Steareth 21) in the remammg water with heat (120° F) and agitation Remove the mix from heat when the surfactants are completely dissolved Next, add the preservatives and the silicone microemulsion to the mix with vigorous agitation
- the final concentrate step involves combining the first and second premixes under vigorous agitation and placing the concentrate in a suitable aluminum can at 7% of total fill with an appropriate mousse valve
- the valve is crimped to the can and a vacuum is applied to deaerate The can is then pressurized with propellant
- the first premix is prepared by adding the Gantrez and PVP/VA to ethanol
- the premix is then neutralized with AMP under vigorous agitation Under continued agitation, add V_ of the water (save remaining V_ for premixing the Silicone Microemulsion, preservatives and surfactants) and the perfume
- the second premix is prepared by dissolving the surfactants (Lauramide DEA, Steareth 21) in the remaining water with heat (120° F) and agitation Remove the mix from heat when the surfactants are completely dissolved Adjust the pH of the Polyquate ⁇ um-47 with AMP to be equal to that of the first neutralized Gantrez premix above Add the pH adjusted Polyquate ⁇ um-47 to the mix with vigorous agitation Next, add the preservatives and the silicone microemulsion to the mix with vigorous agitation
- the final concentrate step involves combining the first and second premixes under vigorous agitation and placmg the concentrate m a suitable aluminum can at 7% of total fill with an approp ⁇ ate mousse valve The valve is crimped to the can and a vacuum is applied to deaerate The can is then pressurized with propellant
- the first premix involves adding the Styleze CC-10 to V_ the water and the ethanol
- the second premix involves dissolving the surfactant (Oleth-20) in the remaining water with heat (120° F) under vigorous agitation Remove the mix from heat when surfactant is completely dissolved Next, add the preservatives, the perfume, and the silicone microemulsion to the mix with vigorous agitation
- the final concentrate step involves combining the first and second premixes under vigorous agitation and placing the concentrate in a suitable aluminum can at 7% of total fill with an appropriate mousse valve
- the valve is c ⁇ mped to the can and a vacuum is applied to deaerate The can is then pressurized with propellant
- Example VII The following is a non-aerosol mousse composition representative of the present invention.
- the first premix is prepared by addmg both polymers PVP/VA and Copolymer 845 to half of the water with agitation and then adding the preservatives.
- the second premix is prepared by addmg the Sodium Cocoyl Isethionate to 40% of the water (heat and vigorous agitation may be required) Combine the Isosteareth-10 and perfume and add to the mix with agitation Next, add the silicone microemulsion, premixed with the remaining (10%) of the water with agitation
- the final concentrate step involves combining the first and second premixes
- Example VIII The following is a Sculpting Spray representative of the present invention: Concentrate composition
- DC- 1845 - Silicone microemulsion from Dow Coming with a particle size of 33 nm, an aniomc/nonionic surfactant system, and a silicone with an internal phase viscosity 77,000 cps
- This product is prepared by combining the alcohol and water (95% of the water) Begm agitation and add the neutralizer (Aminomethyl propanol). Next, add the Ommrez-2000 and mix until completely dissolved Then add the fragrance with agitation and finally add the silicone microemulsion premixed with the remaining water
- This product is prepared by combining the alcohol and water (95% of the water). Begin agitating and add the neutralizer (KOH) Next, add the Amphomer LV-71 and mix until completely dissolved. Add the propylene glycol and glycerin. Finally, under agitation, add the fragrance and the silicone microemulsion (premixed with the remaining water)
- Example X The following is a spray gel composition representative of the present invention.
- This concentrate is prepared by completely dissolving the Polyquate ⁇ um-11 and PVP/VA in the ethanol and the batch water with vigorous agitation Under continued vigorous agitation, add the Diethylene glycol, the Isosteareth-20, the perfume, the Glycerin, and preservatives Next, add the silicone microemulsion to the mix with mild agitation
- Example XI The following is a Non-Aerosol Hairspray composition representative of the present invention
- DC-2-5791 - Silicone microemulsion from Dow Coming with a particle size of 49 nm, an aniomc/nonionic surfactant system, and a silicone with an internal phase visocosity 77,000
- This product is prepared by completely dissolving the Acrylate Copolymer in ethanol with vigorous agitation (there should be no visible white flakes) Under continued vigorous agitation, add the dimethyl my ⁇ stal amine, the Isododecane, and the DIBA While still agitating, slowly add the NaOH It is important to achieve a complete neutralization reaction Next, add the perfume under agitation Then add the water and mix until a uniform mixture results Finally, add the silicone microemulsion to the mix with agitation.
- Example XII The following is an Aerosol Hairspray composition representative of the present mvention
- DC-1845 - Silicone microemulsion from Dow Coming with a particle size of 33 nm, an aniomc/nonionic surfactant system, and a silicone with an internal phase viscosity 70,000 cps
- This concentrate is prepared by adding the AMP to the ethanol with agitation Addmg the methacrylate copolymer to the mix with agitation Next, add the cyclomethicone and perfume to the mix with agitation Then, add the co ⁇ osion inhibitors to the water and add the water to the batch
- the batch concentrate is put into a suitable contamer, with a suitable valve and is pressurized with the propellants
- a premix is prepared by adding the Polyquatern ⁇ um-47 to 1/2 of the water under moderate agitation Next the ammonium lauryl sulfate and Polyacrylate/Polyacrylate copolymer blend are added Preservatives and silicone microemulsion are then added to the mix
- Asecond premix is prepared by adding the hydroxypropyl guar to 1/2 of the water, mixing vigorously until the guar hydrates completely The first and second premixs are then combmed
- a third premix is comprised of propylene glycol, T ⁇ closan, ethanol, and perfume This last ethanol premix is then added to the main mix
- Example XIV The following is a composition for cleansing towellettes representative of the present invention
- the premix is prepare by adding the Chitosomum 2-hydroxyprop ⁇ on ⁇ c acid salt to 60% of the water (180° F) with vigorous agitation Then add the silicone microemulsion (premixed with the remaining water) to the mix with mild agitation Next under continued agitation, add the Ethanol, and the Perfume
- Example XV The following is an Instant Hand Samtizer gel composition representative of the present invention-
- Example XVII The following is a clear acne medication composition representative of the present invention
- DC-2-5932 silicone microemulsion from Dow Coming with a particle size of 24 nm, a cationic surfactant system, and a silicone with an internal phase visocosity 1 ,200 cps
- Example XVIII The following is a Spray-on Suncreen composition representative of the present invention.
- premix 2 combine remaining ingredients; heat as necessary to solubihze components (this may require heated pressure vessel to prevent evaporation of ethanol) Cool the premix to room temperature and combine with premix A
- Aerosol Shaving Cream composition representative of the present mvention.
- DC-2-8194 - Ammosihcone microemulsion from Dow Commg with a particle size of 25 nm, a cationic surfactant system, and a silicone with an internal phase viscosity 4,000 cps.
- the first premix is prepared by addmg the ethanol to V_ of the water (save remaining 'A for premixmg Silicone Microemulsion, preservatives and surfactants) Next, add the Polyquaternium 47 and mix until the Polyquaternium 47 is uniformly dissolved m ethanol/water Then add the Lubrajel Oil. With vigorous agitation, add the perfume and oleyl alcohol.
- the second premix is prepared by dissolving the surfactants (Lauramide DEA, Steareth 21) in the remaining water with heat (120° F) and agitation Remove the mix from heat when the surfactants are completely dissolved Next, add the preservatives and the silicone microemulsion to the mix with vigorous agitation
- the final concetrate step involves combining the first and second premixes under vigorous agitation and placing the concentrate in a suitable aluminum can at 7% of total fill with an appropriate mousse valve The valve is crimped to the can and a vacuum is applied to deaerate The can is then pressurized with propellant
- Example XX The following is a clear 2-m-l Shampoo composition representative of the present invention
- the first premix is prepared by addmg the ethanol to the Polyquaternium 47 and mix until the Polyquaternium 47 is uniformly dissolved m ethanol/water With vigorous agitation, add Dimethyl lauryl amine, the perfume and the Isododecane
- the second premix is prepared by dissolving the surfactants (Lauramide DEA, Steareth 21) in the remaining water with heat (120° F) and agitation Then add the ammonium lauryl sulfate Remove the mix from heat when the surfactants are completely dissolved Next, add the preservatives and the silicone microemulsion to the mix with vigorous agitation
- the final concetrate step involves combining the first and second premixes with agitation
- the following is a shampoo composition for a non-aerosol foamer package representative of the present invention
- the first premix is prepared by adding the Polyquaternium- 11 to half of the water with agitation and then adding the preservatives.
- the second premix is prepared by adding the Sodium Cocoyl Isethionate and Lauryamide DEA to the remaining water (heat and vigorous agitation may be required). Combine the Isosteareth-10 and perfume and add to the mix with agitation. Next, add the silicone microemulsion with mild agitation.
- the final concetrate step involves combining the first and second premixes.
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2000518637A JP2001521880A (en) | 1997-11-05 | 1998-11-02 | Personal care composition |
| BR9815268-8A BR9815268A (en) | 1997-11-05 | 1998-11-02 | Compositions for personal care |
| CA002309509A CA2309509A1 (en) | 1997-11-05 | 1998-11-02 | Personal care compositions |
| EP98949190A EP1028699A1 (en) | 1997-11-05 | 1998-11-02 | Personal care compositions suitable for styling hair |
| AU95555/98A AU9555598A (en) | 1997-11-05 | 1998-11-02 | Personal care compositions |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US08/964,721 | 1997-11-05 | ||
| US08/964,721 US5985294A (en) | 1997-11-05 | 1997-11-05 | Personal care compositions |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1999022698A1 true WO1999022698A1 (en) | 1999-05-14 |
Family
ID=25508890
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB1998/001749 WO1999022698A1 (en) | 1997-11-05 | 1998-11-02 | Personal care compositions |
Country Status (12)
| Country | Link |
|---|---|
| US (1) | US5985294A (en) |
| EP (1) | EP1028699A1 (en) |
| JP (1) | JP2001521880A (en) |
| CN (1) | CN1284852A (en) |
| AR (1) | AR015480A1 (en) |
| AU (1) | AU9555598A (en) |
| BR (1) | BR9815268A (en) |
| CA (1) | CA2309509A1 (en) |
| CO (1) | CO5011106A1 (en) |
| PE (1) | PE127599A1 (en) |
| WO (1) | WO1999022698A1 (en) |
| ZA (1) | ZA9810085B (en) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1999066888A1 (en) * | 1998-06-22 | 1999-12-29 | The Procter & Gamble Company | Hair styling compositions |
| WO2000012053A2 (en) * | 1998-09-01 | 2000-03-09 | Amway Corporation | Makeup compositions and methods of making same |
| WO2001012153A1 (en) * | 1999-08-18 | 2001-02-22 | Colgate-Palmolive Company | Skin cleansing composition with improved stability |
| EP1217985A1 (en) * | 1999-08-16 | 2002-07-03 | Playtex Products, Inc. | Sunscreen lotion or spray composition |
| EP1321127A3 (en) * | 1999-09-16 | 2003-07-02 | L'oreal | Cosmetic composition containing at least one silicone/acrylate copolymer and at least one photoprotectant |
| EP0966248B1 (en) * | 1997-12-31 | 2003-11-12 | L'oreal | Compositions for treating keratinous materials containing a combination of a zwitterion polymer and a water insoluble non-volatile silicon |
| EP1627625A2 (en) * | 2004-08-11 | 2006-02-22 | L'oreal | Cosmetic composition based on a cationic surfactant, a silicone containing either an amine or a vinyl, a fatty alcohol and a propelling agent |
| EP1017359B2 (en) † | 1998-07-25 | 2008-07-09 | Wella Aktiengesellschaft | Hair-setting agent with amphoteric and acidic polymers |
| US8002151B2 (en) | 2004-05-07 | 2011-08-23 | Deb Ip Limited | Method of producing foamed cleanser with suspended particles therein and a dispenser therefore |
| WO2013092073A3 (en) * | 2011-12-22 | 2014-01-30 | Henkel Ag & Co. Kgaa | Product for keratin-containing fibers, containing at least a specific copolymer of the n-vinylpyrrolidone and at least a polymer with structural units derived from maleic acid ester |
| US9718069B2 (en) | 2014-05-12 | 2017-08-01 | Deb Ip Limited | Foam pump |
| US11696886B2 (en) | 2017-05-31 | 2023-07-11 | Shiseido Company, Ltd. | Cosmetic |
Families Citing this family (39)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6180576B1 (en) * | 1998-08-20 | 2001-01-30 | Allan L. Melby | Conditioning shampoo compositions |
| DE69936172T2 (en) * | 1998-09-11 | 2008-01-31 | Rohm And Haas Co. | A process for corrosion inhibition in an aqueous aerosol or foam hair styling composition |
| US6432390B1 (en) * | 1999-03-16 | 2002-08-13 | 220 Laboratories | Low VOC methyl acetate hair sprays |
| CA2300491C (en) * | 1999-03-24 | 2009-12-15 | Unilever Plc | Hair styling compositions |
| US6861397B2 (en) * | 1999-06-23 | 2005-03-01 | The Dial Corporation | Compositions having enhanced deposition of a topically active compound on a surface |
| US6107261A (en) * | 1999-06-23 | 2000-08-22 | The Dial Corporation | Compositions containing a high percent saturation concentration of antibacterial agent |
| US6808701B2 (en) | 2000-03-21 | 2004-10-26 | Johnson & Johnson Consumer Companies, Inc. | Conditioning compositions |
| DE10028524A1 (en) * | 2000-06-08 | 2001-12-20 | Goldwell Gmbh | hair spray |
| FR2823110B1 (en) * | 2001-04-06 | 2004-03-05 | Oreal | PROCESS FOR PERMANENT DEFORMATION OF HAIR USING AMINO SILICONES |
| US20040005285A1 (en) * | 2001-10-03 | 2004-01-08 | The Procter & Gamble Company | Conditioner containing particles |
| JP2005504819A (en) * | 2001-10-03 | 2005-02-17 | ザ プロクター アンド ギャンブル カンパニー | Conditioner containing particles |
| EP1453474A1 (en) * | 2001-10-03 | 2004-09-08 | The Procter & Gamble Company | Composition for improving hair volume |
| CA2459919A1 (en) * | 2001-10-03 | 2003-04-10 | The Procter & Gamble Company | Shampoo containing a cationic polymer and particles |
| US20030086886A1 (en) * | 2001-10-03 | 2003-05-08 | Sanjeev Midha | Hair styling composition containing particles |
| AU2003237505A1 (en) * | 2002-06-18 | 2003-12-31 | The Procter & Gamble Company | Composition containing a cationic polymer with a high charge density and a conditioning agent |
| JP2006511470A (en) * | 2002-09-10 | 2006-04-06 | 高砂香料工業株式会社 | Compositions comprising silicone-in-water emulsions and perfume and hair care preparations comprising such compositions |
| GB0222500D0 (en) * | 2002-09-27 | 2002-11-06 | Unilever Plc | Packaged personal care compositions |
| DE10253591A1 (en) * | 2002-11-15 | 2004-05-27 | Basf Ag | Use of polymers of basic amino group-containing monomers, e.g. N-vinyl-imidazole, as additives for cosmetic, dermatological or pharmaceutical formulations, especially delayed-release salicylic acid preparations |
| BRPI0407373B1 (en) * | 2003-02-12 | 2015-04-22 | Stiefel Res Australia Pty Ltd | Water Resistant Photoprotective Foaming and Foaming Composition |
| BRPI0504779A (en) * | 2004-11-08 | 2006-06-27 | Rohm & Haas | aqueous composition, method of preparation thereof, and aqueous formulation |
| US7585922B2 (en) * | 2005-02-15 | 2009-09-08 | L'oreal, S.A. | Polymer particle dispersion, cosmetic compositions comprising it and cosmetic process using it |
| US7820152B2 (en) * | 2005-05-13 | 2010-10-26 | The Gillette Company | Shave gel composition containing glyceryl acrylate/acrylic acid copolymer |
| US20060257350A1 (en) * | 2005-05-13 | 2006-11-16 | The Gillette Company | Shave composition containing three types of lubricants |
| US8277788B2 (en) | 2005-08-03 | 2012-10-02 | Conopco, Inc. | Quick dispersing hair conditioning composition |
| EP2091512B1 (en) * | 2006-12-21 | 2017-10-11 | The Procter and Gamble Company | Personal care composition comprising a silicone elastomer |
| US20100297041A1 (en) * | 2007-06-15 | 2010-11-25 | The Procter & Gamble Company | Stable dispersions of solid particles in a hydrophobic solvent and methods of preparing the same |
| US20090082472A1 (en) * | 2007-09-25 | 2009-03-26 | Peters Marlin W | Hand sanitizer and method of preparation |
| ES2383253T5 (en) * | 2007-10-30 | 2015-03-06 | Gojo Ind Inc | Hydroalcoholic gels compositions for use with dispensers |
| US20100272657A1 (en) | 2009-04-27 | 2010-10-28 | Akzo Nobel Chemicals International B.V. | Enhanced efficiency of sunscreen compositions |
| AU2010202421B2 (en) * | 2009-06-15 | 2014-05-08 | Gojo Industries, Inc. | Method and compositions for use with gel dispensers |
| US20110272320A1 (en) * | 2010-05-06 | 2011-11-10 | Ali Abdelaziz Alwattari | Aerosol Shave Composition Comprising A Hydrophobical Agent Forming At Least One Microdroplet |
| US10265263B2 (en) * | 2011-09-13 | 2019-04-23 | Lubrizol Advanced Materials, Inc. | Surfactant responsive emulsion polymerized micro-gels |
| WO2013040178A1 (en) * | 2011-09-13 | 2013-03-21 | Lubrizol Advanced Materials, Inc. | Surfactant responsive dispersion polymerized micro-gels |
| BR112014020866A2 (en) | 2012-02-24 | 2019-09-24 | Gojo Ind Inc | microbicidal and sparkling alcoholic compositions |
| KR102131604B1 (en) | 2012-10-11 | 2020-07-09 | 다우 실리콘즈 코포레이션 | Aqueous silicone polyether microemulsions |
| DE102013226048A1 (en) | 2013-12-16 | 2015-06-18 | Henkel Ag & Co. Kgaa | Styling spray with volume effect |
| US10334853B2 (en) | 2016-03-29 | 2019-07-02 | Larada Sciences | Compositions and methods for treating ectoparasite infestation |
| US11628129B2 (en) | 2017-04-04 | 2023-04-18 | Gojo Industries, Inc. | Methods and compounds for increasing virucidal efficacy in hydroalcoholic systems |
| DE102018222037A1 (en) * | 2018-12-18 | 2020-06-18 | Henkel Ag & Co. Kgaa | Composition and method for temporarily deforming keratin fibers |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0468721A1 (en) * | 1990-07-23 | 1992-01-29 | Unilever Plc | Shampoo composition |
| WO1998019654A1 (en) * | 1996-11-04 | 1998-05-14 | The Procter & Gamble Company | Hair mousse composition comprising silicone emulsion |
Family Cites Families (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4584356A (en) * | 1984-11-02 | 1986-04-22 | General Electric Company | Method for making silicone-organic block polymers |
| US4728571A (en) * | 1985-07-19 | 1988-03-01 | Minnesota Mining And Manufacturing Company | Polysiloxane-grafted copolymer release coating sheets and adhesive tapes |
| US4659777A (en) * | 1985-11-27 | 1987-04-21 | Thoratec Laboratories Corp. | Polysiloxane/poly(oxazoline) copolymers |
| US4689289A (en) * | 1986-04-30 | 1987-08-25 | General Electric Company | Block polymer compositions |
| US4693935A (en) * | 1986-05-19 | 1987-09-15 | Minnesota Mining And Manufacturing Company | Polysiloxane-grafted copolymer pressure sensitive adhesive composition and sheet materials coated therewith |
| US4733677A (en) * | 1986-11-04 | 1988-03-29 | Dow Corning Corporation | Hair fixative composition containing cationic organic polymer and polydiorganosiloxane microemulsions |
| JPS63130512A (en) * | 1986-11-18 | 1988-06-02 | Toray Silicone Co Ltd | Cosmetic |
| US4871529A (en) * | 1988-06-29 | 1989-10-03 | S. C. Johnson & Son, Inc. | Autophobic silicone copolyols in hairspray compositions |
| DK0412704T3 (en) * | 1989-08-07 | 1999-11-01 | Procter & Gamble | Hair conditioning and styling compositions |
| US5658557A (en) * | 1989-08-07 | 1997-08-19 | The Procter & Gamble Company | Hair care compositions comprising silicone-containing copolymers |
| US5032460A (en) * | 1989-08-14 | 1991-07-16 | Minnesota Mining And Manufacturing Company | Method of making vinyl-silicone copolymers using mercapto functional silicone chain-transfer agents and release coatings made therewith |
| US5017221A (en) * | 1989-12-05 | 1991-05-21 | Dow Corning Corporation | Polymethylalkylsiloxane emulsions and methods |
| CA2041599A1 (en) * | 1990-06-01 | 1991-12-02 | Michael Gee | Method for making polysiloxane emulsions |
| CA2043282A1 (en) * | 1990-06-07 | 1991-12-08 | Ronald P. Gee | Process for making polysiloxane emulsions |
| US5085859A (en) * | 1990-07-06 | 1992-02-04 | Dow Corning Corporation | Hair fixatives |
| US5244598A (en) * | 1991-09-13 | 1993-09-14 | General Electric Company | Method of preparing amine functional silicone microemulsions |
| GB9204509D0 (en) * | 1992-03-02 | 1992-04-15 | Unilever Plc | Hair care composition |
| US5468477A (en) * | 1992-05-12 | 1995-11-21 | Minnesota Mining And Manufacturing Company | Vinyl-silicone polymers in cosmetics and personal care products |
| EP0582152B1 (en) * | 1992-07-28 | 2003-04-16 | Mitsubishi Chemical Corporation | A hair cosmetic composition |
| US5393452A (en) * | 1992-11-09 | 1995-02-28 | General Electric Company | 2 in 1 shampoo system and conditioner comprising a silicon-polyether copolymer |
| DE4338421A1 (en) * | 1993-11-10 | 1995-05-11 | Wacker Chemie Gmbh | Graft copolymers of organopolysiloxanes as free-radical macroinitiators |
| US5578298A (en) * | 1994-05-27 | 1996-11-26 | General Electric Company | Microemulsions for high viscosity amino silicone fluids and gums and their preparation |
| US5534265A (en) * | 1994-08-26 | 1996-07-09 | The Procter & Gamble Company | Thickened nonabrasive personal cleansing compositions |
| FR2740032B1 (en) * | 1995-10-18 | 1997-11-21 | Oreal | WATER-BASED LACQUER FOR THE TREATMENT OF KERATINIC MATERIALS, PACKAGED IN AN AEROSOL DEVICE, INCLUDING AT LEAST ONE SILICONE POLYMER GRAFT AND APPLICATIONS |
-
1997
- 1997-11-05 US US08/964,721 patent/US5985294A/en not_active Expired - Fee Related
-
1998
- 1998-11-02 BR BR9815268-8A patent/BR9815268A/en not_active Application Discontinuation
- 1998-11-02 WO PCT/IB1998/001749 patent/WO1999022698A1/en not_active Application Discontinuation
- 1998-11-02 JP JP2000518637A patent/JP2001521880A/en not_active Abandoned
- 1998-11-02 CN CN98812951A patent/CN1284852A/en active Pending
- 1998-11-02 AU AU95555/98A patent/AU9555598A/en not_active Abandoned
- 1998-11-02 EP EP98949190A patent/EP1028699A1/en not_active Withdrawn
- 1998-11-02 CA CA002309509A patent/CA2309509A1/en not_active Abandoned
- 1998-11-04 CO CO98064815A patent/CO5011106A1/en unknown
- 1998-11-04 ZA ZA9810085A patent/ZA9810085B/en unknown
- 1998-11-05 PE PE1998001050A patent/PE127599A1/en not_active Application Discontinuation
- 1998-11-05 AR ARP980105600A patent/AR015480A1/en not_active Application Discontinuation
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0468721A1 (en) * | 1990-07-23 | 1992-01-29 | Unilever Plc | Shampoo composition |
| WO1998019654A1 (en) * | 1996-11-04 | 1998-05-14 | The Procter & Gamble Company | Hair mousse composition comprising silicone emulsion |
Cited By (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0966248B1 (en) * | 1997-12-31 | 2003-11-12 | L'oreal | Compositions for treating keratinous materials containing a combination of a zwitterion polymer and a water insoluble non-volatile silicon |
| WO1999066888A1 (en) * | 1998-06-22 | 1999-12-29 | The Procter & Gamble Company | Hair styling compositions |
| EP1017359B2 (en) † | 1998-07-25 | 2008-07-09 | Wella Aktiengesellschaft | Hair-setting agent with amphoteric and acidic polymers |
| WO2000012053A2 (en) * | 1998-09-01 | 2000-03-09 | Amway Corporation | Makeup compositions and methods of making same |
| WO2000012053A3 (en) * | 1998-09-01 | 2000-06-29 | Amway Corp | Makeup compositions and methods of making same |
| US6132739A (en) * | 1998-09-01 | 2000-10-17 | Amway Corporation | Makeup compositions and methods of making same |
| EP1217985A1 (en) * | 1999-08-16 | 2002-07-03 | Playtex Products, Inc. | Sunscreen lotion or spray composition |
| EP1217985A4 (en) * | 1999-08-16 | 2003-07-30 | Playtex Products Inc | Sunscreen lotion or spray composition |
| WO2001012153A1 (en) * | 1999-08-18 | 2001-02-22 | Colgate-Palmolive Company | Skin cleansing composition with improved stability |
| AU774800B2 (en) * | 1999-08-18 | 2004-07-08 | Colgate-Palmolive Company, The | Skin cleansing composition with improved stability |
| EP1321127A3 (en) * | 1999-09-16 | 2003-07-02 | L'oreal | Cosmetic composition containing at least one silicone/acrylate copolymer and at least one photoprotectant |
| US8281958B2 (en) | 2004-05-07 | 2012-10-09 | Deb Ip Limited | Method of producing foamed cleanser with suspended particles therein and a dispenser therefore |
| US8002151B2 (en) | 2004-05-07 | 2011-08-23 | Deb Ip Limited | Method of producing foamed cleanser with suspended particles therein and a dispenser therefore |
| AU2005239772B2 (en) * | 2004-05-07 | 2011-09-15 | Deb Ip Limited | Foamed cleanser with suspended particles |
| US10736824B2 (en) | 2004-05-07 | 2020-08-11 | Deb Ip Limited | Foamed cleanser with suspended particles |
| EP1627625A3 (en) * | 2004-08-11 | 2008-04-23 | L'oreal | Cosmetic composition based on a cationic surfactant, a silicone containing either an amine or a vinyl, a fatty alcohol and a propelling agent |
| EP1627625A2 (en) * | 2004-08-11 | 2006-02-22 | L'oreal | Cosmetic composition based on a cationic surfactant, a silicone containing either an amine or a vinyl, a fatty alcohol and a propelling agent |
| WO2013092073A3 (en) * | 2011-12-22 | 2014-01-30 | Henkel Ag & Co. Kgaa | Product for keratin-containing fibers, containing at least a specific copolymer of the n-vinylpyrrolidone and at least a polymer with structural units derived from maleic acid ester |
| US9132079B2 (en) | 2011-12-22 | 2015-09-15 | Henkel Ag & Co. Kgaa | Product for keratin-containing fibers, containing at least a specific copolymer of the N-vinylpyrrolidone and at least a polymer with structural units derived from maleic acid ester |
| US9718069B2 (en) | 2014-05-12 | 2017-08-01 | Deb Ip Limited | Foam pump |
| US11696886B2 (en) | 2017-05-31 | 2023-07-11 | Shiseido Company, Ltd. | Cosmetic |
Also Published As
| Publication number | Publication date |
|---|---|
| CA2309509A1 (en) | 1999-05-14 |
| BR9815268A (en) | 2001-05-15 |
| EP1028699A1 (en) | 2000-08-23 |
| JP2001521880A (en) | 2001-11-13 |
| PE127599A1 (en) | 2000-02-17 |
| ZA9810085B (en) | 1999-05-05 |
| AR015480A1 (en) | 2001-05-02 |
| US5985294A (en) | 1999-11-16 |
| CO5011106A1 (en) | 2001-02-28 |
| AU9555598A (en) | 1999-05-24 |
| CN1284852A (en) | 2001-02-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5985294A (en) | Personal care compositions | |
| US6149898A (en) | Hair styling compositions containing silicone microemulsions and cationic non-polymeric liquids | |
| US5972356A (en) | Personal care compositions | |
| US5965115A (en) | Personal care compositions | |
| US5985295A (en) | Personal care compositions | |
| EP1028704B1 (en) | Personal care compositions | |
| US5997886A (en) | Personal care compositions | |
| AU694441B2 (en) | Hair styling compositions containing a silicone grafted polymer and low level of a volatile hydrocarbon solvent | |
| US5968495A (en) | Personal care compositions | |
| WO2002102335A1 (en) | Hair styling compositions | |
| US20030086886A1 (en) | Hair styling composition containing particles | |
| AU705329B2 (en) | Hair cosmetic compositions | |
| WO2002011684A2 (en) | Water-in-oil emulsion compositions comprising polyalkylene glycol styling agents | |
| JPH06509808A (en) | Hairspray composition containing fluorosurfactant | |
| MXPA00012424A (en) | Hair styling compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 98812951.5 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AL AM AT AT AU AZ BA BB BG BR BY CA CH CN CU CZ CZ DE DE DK DK EE EE ES FI FI GB GE GH GM HR HU ID IL IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SK SL TJ TM TR TT UA UG UZ VN YU ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW SD SZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| ENP | Entry into the national phase |
Ref document number: 2309509 Country of ref document: CA Ref document number: 2309509 Country of ref document: CA Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 95555/98 Country of ref document: AU |
|
| NENP | Non-entry into the national phase |
Ref country code: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1998949190 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1998949190 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1998949190 Country of ref document: EP |